![]() In my field, many things that cause the average man-on-the-street to get a bit squeamish or squicked are rather commonplace. My own studies include two types of bacteria that are carried rectally in humans (and other animals), so I spend an absurd amount of time thinking about, well, shit, and the lifeforms that inhabit it and collectively make up our normal gut flora. The vast majority of these species don't harm us at all, and many are even beneficial: priming our immune system; assisting in digestion; and filling niches that could be colonized by their nastier bacterial brethren.
In my field, many things that cause the average man-on-the-street to get a bit squeamish or squicked are rather commonplace. My own studies include two types of bacteria that are carried rectally in humans (and other animals), so I spend an absurd amount of time thinking about, well, shit, and the lifeforms that inhabit it and collectively make up our normal gut flora. The vast majority of these species don't harm us at all, and many are even beneficial: priming our immune system; assisting in digestion; and filling niches that could be colonized by their nastier bacterial brethren.
It's typically when there's some disturbance in these flora that bad things happen. For example, you may ingest food contaminated with a foreign bacterial strain that may transiently colonize your intestines, resulting in cramping and diarrhea. Typically these infections are self-limited and your normal flora "resets itself" after a short time, but some pathogenic bacteria have a propensity for making themselves at home in your gut. How to get rid of these nasty invaders then? Antibiotics are one option, but they also kill your regular bacteria, potentially making the problem worse (especially if the nasty invader happens to be resistant to many antibiotics). There has been a large increase in the use of probiotics--formulations designed to add beneficial bacteria to your gut. However, these have largely not been rigorously tested or regulated, so it's unsure how well they actually work.
What if, instead of re-constitituing healthy gut flora one species at a time, you could simply take the entire fecal contents from a healthy person and use it to re-colonize your own gut--in other words, undergo a fecal transplant? Yes, it's like probiotics on steroids: getting an infusion of someone else's gut flora in order to re-establish a healthy gut ecology of your own, and squeeze out some potentially harmful organisms along the way. A recent story discusses this treatment for patients suffering Clostridium difficile infections in Scotland, but it's actually not brand-new, and has already surfaced in the peer-reviewed literature. More after the jump...
First, a quick review of C. difficile, which is a spore-forming bacterium carried asymptomatically by a small percentage of us. This species has become a problem in recent years due to both the emergence of a new, more virulent strain, and apparently due to an increase in use of a certain class of antibiotics, the fluoroquinolones.
These antibiotics are termed "broad spectrum:" they kill a number of different species of bacteria in one fell swoop. This is good for the clinician, because it means they can start treatment quickly, before culture results are even back from the lab. However, it may be bad for the patient in the long term because it means that the antibiotic regimen will kill not only the bacterium causing the disease, but also will wipe out many beneficial organisms in and on the body. The result can be a disturbance in the ecology of one's normal flora, setting the stage for an invader such as C. difficile to come in and set up shop--and once it's there, it's notoriously difficult to get rid of. And once it's there and causing a symptomatic infection, it can be hell to deal with, resulting in copious and sometimes frequent diarrhea, and occasionally causes a more serious and painful condition called colitis (inflammation of the colon). Additional antibiotics can eliminate C. difficile, but they don't work for all patients, and infection can result in miserable symptoms. Thus, some have turned to the fecal transplant as a last-ditch effort to cure themselves of the infection.
This procedure was described in a 2003 Clinical Infectious Diseases paper, documenting 19 patients who'd undergone a fecal transplant between 1994 and 2002. Donor feces are provided to the patient via a nasogastric tube as depicted in the picture to the right. 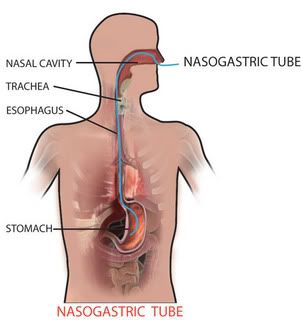 First, of course, donor stool must be procured. When possible, they used donor stool from someone the recipient would be in contact with anyway--a spouse or other household member, preferably. A fresh sample is obtained and then, um, processed. The authors describe their methods (emphasis mine):
First, of course, donor stool must be procured. When possible, they used donor stool from someone the recipient would be in contact with anyway--a spouse or other household member, preferably. A fresh sample is obtained and then, um, processed. The authors describe their methods (emphasis mine):
Select a stool specimen (preferably a soft specimen) with a weight of 30 g or a volume of 2 cm^3. Add 50-70 mL of sterile 0.9 N NaCl to the stool sample and homogenize with a household blender. Initially use the low setting until the sample breaks up; then, advance the speed gradually to the highest setting. Continue for 2-4 min until the sample is smooth. Filter the suspension using a paper coffee filter. Allow adequate time for slow filtration to come to an end. Refilter the suspension, again using a paper coffee filter. As before, allow adequate time for slow filtration.
25 mL of the suspension is then transferred to the recipient, who's already been prepared for the transplant via treatment with vancomycin (to kill off as much existing C. difficile as possible) and omeprazole (Prilosec, to decrease stomach acid production). The tube is then flushed with a salt solution and removed, and the recipient is free to go. (They were followed up either via phone or return visits to the clinic).
Was it worth it? It appeared to be a fairly successful procedure:
After the stool transplantation, 14 of the 16 surviving patients submitted a total of 20 stool samples that were tested for C. difficile toxin. Patients 3 and 11 did not submit stool samples after undergoing stool transplantation. A telephone followâup conversation with patient 3 and a review of the clinical record for patient 11 verified that neither patient had experienced a recurrence of diarrhea after the stool transplantation. Both patients remained free of diarrhea during the 90âday followâup period.
One additional patient did develop diarrhea due to C. difficile, and was given an additional course of vancomycin (after which he was fine). Additionally, "all surviving patients reported that bowel habits returned to the functional pattern that had preceded their first episode of C. difficile colitis."
The authors acknowledge that additional research needs to be done (including clinical trials). A limitation exists in the design: one can't be certain if it was the vancomycin treatment or the fecal transplant that led to the resolution of symptoms (although the former seems unlikely, since recipients were all patients who had previously received vancomycin treatment). Additionally, there's obviously a lot of "ick" factor that needs to be overcome. There are also several logistic hurdles, such as storing and transporting feces in some cases:
"I had to collect stool samples for five days prior to our leaving Toronto, and I collected it in an ice cream container and kept it in the fridge," said [donor] Sinukoff.
She had to then fly the samples to Calgary so that [Calgary physician Dr. Tom] Louie could transplant it into her sister -- a process that involved getting the sample through airport security.
"My biggest fear was that my samples were not allowed to be frozen, so I had to take them as carry-on luggage in the airplane and I was terrified that I was going to be asked to have my luggage searched," she said.
As the article quips, in the annals of medical history, this method has the potential to be one of the most effective, but also most stomach-churning.
Reference
Aas, J. et al. 2003. Recurrent Clostridium difficile Colitis: Case Series Involving 18 Patients Treated with Donor Stool Administered via a Nasogastric Tube. CID. 36:580-585. Link.
Image from http://www.disaboom.com/getfile/6e427b25-7801-4474-b794-fe8e5aab179c/Na…
- Log in to post comments

Thanx
"take the entire fecal contents from a healthy person and use it to re-colonize your own gut"
One problem is, of course, what is "healthy"? Does my 'healthy' mean the same, in terms of gut ecology, as the (apparently`)'healthy' of my 'donor'? I suppose sharing an overall environment, including diet, would help. But how similar is body flora of presumibly 'healthy' people in the same environment? What research I have seen seems to indicate a large variation, ie there are few things everyone seems to have in abundance, then there is all the rest,highly variable, which usually where the problems are. I might just be switching (or adding) someone elses problems for mine. Seems risky until we know a bit more about it.
It still varies, and sure, there are risks. They note in the methods they screened for several other potential enteric pathogens, as well as parasites and ova, and did blood tests for a number of blood-borne pathogens that could theoretically be introduced during such a procedure as well. It's certainly not something you'd want to do for anything minor, but C. difficile infections can be chronic and very tough to get rid of, and keep people practically confined to their bathrooms in the worst cases. These patients all had been treated for it previously, and treatment failed--leaving few other options.
So... Finally a use for which creationists appear to be specially designed! If I read this correctly could one Michael Behe could potentially treat an entire continent?
This pinged an old memory. I seem to recall in William Nolan's book "Making of a Surgeon" (which I think I read in 1971) his account of a colleague using a similar technique to treat a case of staph infection at Bellevue, I think in the late 1950's.
Apparently the "healthy stool" was introduced by mouth, mixed in with chocolate milk! (I will let that thought linger without comment.) Interestingly, when the resident was called on the carpet for using an untested technique, he claimed to have used a procedure very similar to the one you describe (filtering, sterile saline, etc,) probably hoping to diminish the ick factor.
Obviously the story was vivid enough to hang in my mind all these decades. I can't help wondering if this meme has been bouncing around the medical world for many years.
I've been wondering about this as apparently it's sometimes used to treat Crohn's disease and I know a few people who suffer from that.
Then there's the "give your immune system something to do" approach, which gives Crohn's patients pig parasites so their immune system works on them and leaves off attacking the intestine. The parasites don't multiply in the human gut so the patient takes a pill of parasite eggs every 2 or 3 months.
It sounds yucky but it has to be better than having your intestine yanked out.
Sorry, that should be every two or three weeks.
They laughed at my theories at the university. They said my research on poop transplantation trampled in domains Man was not meant to tread. They said I was mad! Mad, I tell you! Well who's laughing now? Bwa-ha-ha-ha-haaa!
There are too many jokes that could be made here...
"If God had meant for us to transplant poo, He'd have made it harder to donate!"
"So Mr. Hanky now has a new occupation?"
Seriously though, this is quite cool. Can't wait to see whether this catches on.
Very interesting! I, for one, am not so squeamish or "squick"-able as the average person.
Why were the bacteria introduced to the stomach? It seems more direct and less revolting to introduce them to the colon, and less prone to pH-based eradication of the desired organisms.
(The strain of C.diff that's been on the rise lately is surprisingly horrible and persistent, and I knew some patients who had previously had perfectly adequate bowel control who were repeatedly incontinent after the organism took hold. That's a huge problem for seniors who want to go home to live independently after a stint in postoperative rehab or a brief hospitalization.)
This reminds of advice I found in the literature concerning rabbit husbandry. When a rabbit's digestive system shuts down after a prolongued period without eating, an easy way of relaunching its system was to feed it pellets from a healthy rabbit.
I know, it's a rabbit not a human and their digestive system depends on the continued ingestion of caecal(?) pellets.
If this had to be refrigerated, I think I would be buying an extra refrigerater! I'm so squeamish I can't even stand to keep opened dog food in my fridge. My mother did that when I was a kid and it grossed me out. I'm not sure why.
It seems the Canadian group is doing it the other way--via the colon in an enema-based inoculation. However, the CID authors say in the discussion re: the nasogastric tube method:
Hey, as a solid organ transplant patient I can say the ick factor may be higher, but the procedure and recovery sounds much much better.
Isn't there some statistic of likelihood regarding the amount of shit each person has already consumed, sans ng tube?
This will be an interesting treatment to follow.
Of course, the other question is whether it's possible to develop a more effective culture of bacterium in the lab and use that instead of a fecal transplant?
Then again, I suppose all the nastiness associated with feces comes from the bacteria anyways. =/
A man is visiting his friend when he walks into the kitchen to find him at the counter eating a bowl of poop. Aghast he exclaims, "What are you doing that for!?"
His friend looks at him and says, "I don't know, I've just been feeling like shit all day."
Austin Powers can get recolonized too:
I wonder if the legendary flatulence was passed along in the sample.
Yuck indeed.
But I guess it's no worse than "I'm gonna slice open your body and cut away the bad bits". [No offense to the surgeons out there.]
A suppository would seem a bit less, umm, icky, but I guess less effective at colonization.
Hey, I made a joke, "colonization", gettit, har har.
Potentially, but recall that we can only culture a small part of our normal gut flora--and we have only the most basic notions about the overall ecology of intestinal bacteria and how it's maintained. That's why the "whole poo" transplant, in theory, would be more successful--you're sending along the whole shebang, so you eliminate some of the problems with probiotics and their frequent inability to establish themselves as part of the pre-existing gut ecology.
Tara Smith said:
Minus Hepatitis A and the miscellaneous intestinal parasites
hopefully......I hope the donors are screened appropriately...
Maybe some kind of colonic irrigation with the appropiate
specimen would be more acceptable?
Eat shit and... live?
As a medical student I always used to joke that the specialty of coprology should exist. Now it seems that it does.
Way back in the early 1980's I read a medical lab report on the use of 'normal fecal enemas'. The ick factor was high back then too, but so was the cost of vanco... By the way, the specialty of coprology belongs to the lab and paleontology. Has anyone else noticed how much those hydration systems bikers and hikers use resemble enema bags?
Merry Christmas, Tara!
So, homophobes keep dwelling on the "unsanitary" "disgusting" et cetera sexual habits of goes and talking about fecal matter getting on the sex partner's penis and spreading diseases.
Doesn't this suggest that frequent anal sex as the passive partner with multiple partners might actually increase your chances of maintaining a healthy bacterial ecosystem?
What a nonsense entry. Blogging on peer-reviewed research, aren't you? Well, as far as I can see, it's at least a very cute illustration of the fact that peer review is nothing but inbred science. People who believe shit validate other people's shit.
You know, Tara, the day you understand that flora disturbance is not a cause but a symptom, that day you'll be set on your way to something relevant. Maybe.
That was a great post! It makes a lot of sense to...although you'll excuse me if I stick with my daily bowl of yogurt rather than shooting shit up my nose. I'll save that for when I get desperate!
"and homogenize with a household blender. Initially use the low setting until the sample breaks up; then, advance the speed gradually to the highest setting. "
remind me to not have a smoothie at that dude's place
Several times today I said to myself: "It's a joke, of course, it's April's fool day!" Several times today I found out that, no, it's not the first of April. Scientists really imagined and performed this experience. With real donors. With real patients. This thing has entered the anals of peer reviewed research.
Man, am I proud that I had already recognized, before reading this fecal transplant thing, all by litte self and before the herd nerds started to wake up and smell something's terribly wrong, the peer reviewed evidence based medicine for what it is: bullshit.
Herd nerds waking up, you ask me? Of course, now the shit transfer practice has hit the fan, people cannot fail to finally wake up and reognize the insanity of modern "Post Louis Pasteur" medicine.
10 years ago I suffered with a bout of C. difficile and believe me, I'd gladly have eaten shit with a spoon if it would have given me relief from the never-ending misery I was enduring. The infection is horrible. Look at it this way: Eat shit OR die. Sorry for the 'gross'factor here, but when you're suffering from this disorder, you wish you were dead, just to relieve your misery.
"What a nonsense entry. Blogging on peer-reviewed research, aren't you? Well, as far as I can see, it's at least a very cute illustration of the fact that peer review is nothing but inbred science. People who believe shit validate other people's shit."
Could you, like, back that up with something, like, say, evidence or stuff? You know, anyone can call anything shit. Are you saying that the people who got better after the treatment when nothing had helped them before didn't actually get better?
"Of course, now the shit transfer practice has hit the fan, people cannot fail to finally wake up"
How so? As far as I can tell, your point here is "It sounds really weird, so that proofs that it doesn't make sense." Interesting line of argument from people who take pride in being unconventional rather than "herd-following".
BTW, do I have to remind you that various forms of "alternative medicine" have been big on urine treatments for quite a while?
"and reognize the insanity of modern "Post Louis Pasteur" medicine."
Wich is why life expectancies went down so radically since that was introduced.
As to the "nonsense entry" bit - jspreen, it mystifies me that someone who doesn't believe that microorganisms cause disease so regularly reads a blog about disease written by an infectious disease epidemiologist. You think *every* entry is nonsense. I am sure Dr. Smith takes it as read, so why bother saying so?
Great stuff, Tara. I wish I'd noticed your post before making a casual reference to fecal enemas in my latest post; I've just added an update. It's worth noting that the 2003 case series you referred to was not the first published report of doctors re-seeding a colon to treat C-diff. In 2002, I wrote a cover story for Science News that lead with a case report of such a treatment. In that case, in contrast to the study you blogged about, the doc went in the back door, so to speak. He used a sigmoidoscope, if I recall, to insert of bit of fecal matter from his patient's husband into the lower colon of his patient. (A creative solution to a novel medical challenge, I thought.) By the time I interviewed the patient, her husband had died, but some of his commensals probably lived on in her GI tract.
No mystery here. Most people prefer to hang around with their peers, it's so much easier. But I got a bit tired lately of sharing my ideas with people who already agree and I gave it a try in this snake pit. And yes, I think it's a lot more effective. Mainly to give a chance to the heirs following main stream who never ever heard the name Ryke Geerd Hamer, and will never ever read about the Germanic New Medicine in main stream media or peer reviewed evidence based medicine research columns.
You know how it goes.
You read about something totally new and way off stream. First you think "bullshit". But as times passes by and you read about it more often here and there, the idea eventually finds its way into the brain cells of even the nerdiest.
Mark my words, jen_m. Ryke Geerd Hamer, Germanic New Medicine. In some time, when mankind has become intelligent, GNM will be teh thing. At that same time, todays practices of chemo poisoning, radiation burning and, why not, shit transplanation, etc., will be a source of a mighty lot of thigh slapping.
The increase of life expectancy has little to do with medicine. Modern medicine is surfing on the wave of wealth in the Western contries and, were it not for the absence of life threatening poverty, famine, war or whatever, or scientific medicine would long since have started to decrease life expectancy. Don't forget, Raphael, people who for instance survive a cancer after chemo survived not because but in spite of their treatment. Idem dito for Aids and wonderfull life saving killer drugs like ARVs.
But maybe you're right and I should really stop ranting around here. The world is getting overcrowded and, as I wrote before: Anybody, dumb enough to believe that drugs that make you sick from the first moment you start taking them are good for one's health, should be allowed full access to health-care facilities.
And here I would have thought this would be right up jspreen's alley: all natural, no "toxic" drugs, restoring the body's normal balance.
I note that you didn't answer my main question, so I'll repeat it: Could you, like, back that up with something, like, say, evidence or stuff?
The first thing you said here is that this entry is nonsense. What evidence or compelling arguments do you have for that claim?
"peer reviewed evidence based medicine research columns." So, do I get this right- you yourself admit that peer review is about evidence? Are you admitting that the evidence goes against you?
"You know how it goes.
You read about something totally new and way off stream. First you think "bullshit". "
Do you really not realise how well this describes your first posts in this thread? How much sense does it make for you to assume as a matter of course that something is wrong if it first looks like bullshit to you, but expect others to take exactly the opposite approach to the things you say?
"But as times passes by and you read about it more often here and there, the idea eventually finds its way into the brain cells"
Sometimes, somretimes not. The idea people can live of nothing but air, for instance, still hasn't found a way into my brain cells. (Neither has the idea that Britney Spears makes good music, for that matter.) You know, for the more rational and sane ones among us, wether an idea convinces us depends on wether it has such funny things as "evidence" and "compelling arguments" in its favor.
"Mark my words, jen_m. Ryke Geerd Hamer, Germanic New Medicine. In some time, when mankind has become intelligent, GNM will be teh thing."
So you think mankind will embrace some kind of semi-nazism? That implies that most members of mankind are inferior? Why should mankind do this?
Not to mention that you haven't given any compelling reason to believe that being intelligent implies agreeing with you.
" At that same time, todays practices of chemo poisoning, radiation burning and, why not, shit transplanation, etc., will be a source of a mighty lot of thigh slapping."
Again, could you, like, back that up with something, like, say, evidence or stuff? And while we're at it- by what date do you think will that be so, and what are you willing to bet?
"The increase of life expectancy has little to do with medicine. Modern medicine is surfing on the wave of wealth in the Western contries and, were it not for the absence of life threatening poverty, famine, war or whatever, or scientific medicine would long since have started to decrease life expectancy."
Once again- Evidence? Compelling arguments?
"Don't forget, Raphael, people who for instance survive a cancer after chemo survived not because but in spite of their treatment. Idem dito for Aids and wonderfull life saving killer drugs like ARVs."
Again, what evidence do you have for that? And anyway, what share of medicine do these treatments form? What about people who got bitten by rabid dogs, got vaccinated, and survived? And others, who (since long before modern medicine) got bitten by rabid dogs and died? And people who had an appendictis, got their appendix removed, and survived, and others who got appendictis, didn't get their appendix removed, and died? What about people who survived physical injuries that would have killed them 200, 100, 50, or even 20 years ago? What about people who had a headache, took pills, and were relieved? Talking of pills- what about the Pill? Are you saying that it doesn't have any effect?
"But maybe you're right and I should really stop ranting around here." I don't think so- it might be fun if you tried to argue for your opinions, instead of simply asserting them.
Out of curiosity, do we know how the intestines acquire this colony? Does a newborn have a colony in place at birth, or is it something the child acquires at a later date? If the latter, is that acquisition from mother's milk or does this not begin until solid food is introduced?
There must be a difference between colonies across the human spectrum - otherwise visits to Mexico would not be so fraught with danger of intestinal distress. As a former developing world road warrior, I employed the Bedouin trick of eating raw onions from local sources. This was usually successful when combined with usual precautions.
I recall reading in an article on biofilms that one of the big but rarely-discussed issues among colonoscopists is how to handle the cleaning of the instrument, since it has a hollow center. Apparently there's no known way to really sterilize a colonoscope, given how successfully and rapidly biofilms form (because even if you kill off all but one of the bacteria, then there it is surrounded by the perfect growth medium, the bodies of its peers, and off it goes again).
One gathers they basically figure what they're doing is more important than the risks, or perhaps there's no way to document the risks if any of spreading organisms this way.
Where does a bacterium sit?
Everywhere it wants to.
Onkel Bob, from what I've been able to gather with a quick look at some reviews on the subject, the colonization begins during birth, with the rupture of the membranes and the passage through the vaginal canal (which is colonized by its own microflora and often contaminated with intestinal contents during labor), and continues during nursing, when the infant is exposed to skin and environmental bacteria. Apparently formula-fed infants have gut microflora more similar to adults' than their breastfed peers.
Populations of gut bacteria apparently change in a characteristic developmental manner as infants mature and are exposed to different sources of bacteria, too.
On a side note, I'd always wondered why, given that the human gut usually slows during sustained physical stress (sympathetic ANS arousal), women so often defecated during the strain of childbirth (i.e. they still had stool bulk in the rectum despite many hours of physical effort, unlike, say, marathon runners.) I imagine that perhaps if very early colonization with maternal gut symbionts is protective, it might be that the presence of such materials furnishes a survival benefit that merits continued gut activity. (Or it might just be that for the very long effort of birth, it's worth processing whatever calories are already onboard. I shouldn't get too carried away with my speculations!)
A lot of questions, Raphael. A pity I can't answer you. Too sure you won't listen to my answers, really. Almost nobody can, especially those who swallowed the little Hitler hate campaign against Hamer hook, line and sinker.
Well, OK, I'll give in and answer this one:
Easy. What about people who had a headache, took nothing, and were relieved?
My crowd outnumbers yours, Raphael. A thousand times.
A lot of questions, Raphael. A pity I can't answer you. Too sure you won't listen to my answers, really. Almost nobody can, especially those who swallowed the little Hitler hate campaign against Hamer hook, line and sinker.
Well, OK, I'll give in and answer this one:
Easy:
What about people who had a headache, took nothing, and were relieved?
And:
What about people who had a headache, took pils, and found no relieve?
My crowd outnumbers yours, Raphael. A thousand times. What does that mean? Simply that your kind of questioning doesn't lead anywhere.
Thank you Jen M. Interesting note on bottle fed infants. I'm guessing that the gastric/stomach acids and enzymes are not as highly developed and allow more flora to pass through. I'm not a father (and pushing 50 I hope not to be one!) so I have limited experience with children.
Uncle Bob, from the mother presumably.
I remember reading somewhere a theory that efficient herbivory
(requiring specialized gut bacteria) in evolution only developed together with child care because previously there was no reliable
way to "infect" the babies.
So, basically, you have perfectly fine answers for my questions, but you're not going to tell me about them? Oh, purr-lease. Honestly, do you expect *anyone* to buy that?
Anyway,
"Well, OK, I'll give in and answer this one:
What about people who had a headache, took pills, and were relieved?
Easy:
What about people who had a headache, took nothing, and were relieved?
And:
What about people who had a headache, took pils, and found no relieve?"
None claims that modern mainstream medicine always works, so cases where it doesn't don't refute anything. You, on the other hand, seem to claim that it *never* works, so any single case where it does refutes your beliefs.
"My crowd outnumbers yours, Raphael. A thousand times."
And where's the evidence or comelling arguments for that?
"What does that mean? Simply that your kind of questioning doesn't lead anywhere."
My kind of questioning is basically asking for evidence or compelling arguments. What do you mean when you say that it doesn't lead anywhere? That you don't have evidence or compelling arguments?
While we're at it- don't you think there's a tiny bit of a contradiction between
"Too sure you won't listen to my answers, really. Almost nobody can,"
and
"My crowd outnumbers yours, Raphael. A thousand times."?
Wich one is it?
Another thing I noticed: You talked here about "the insanity of modern "Post Louis Pasteur" medicine." However, judging from your writings, GNM seems to imply that any kind of medicine that relies to a good deal on prescribing some kind of medication or another is wrong- wich would mean, pretty much any kind of medicine the human race ever came up with, pre- or post-Pasteur, mainstream or alternative, modern or ancient. What's supposed to be so particularly insane about "modern "Post Louis Pasteur" medicine", then?
(To everyone else: Sorry, I'm kind of enjoying this at the moment.)
May be some time before this hist the supermarket shelves - although the marketing possibilities of using celebrity 'donors' would be enormous ; )
A small trial of fecal enemas for ulcerative colitis was also highly successful. All 6 UC patients who underwent fecal enemas from healthy donors went into long remissions (in fact it's tempting to say "cured", since UC relapsed in none of them over years). Published in J. Clinical Gastro 2003: http://www.cdd.com.au/pdf/UC%20bacteriotherapy.pdf
Raphael, it just ends in frustration if you think in any way your obvious questions will persuade it to think scientifically.
My, this jspreen person is rather annoying.
If one is using the Firefox browser, and one wishes to not have to wade through the jspreen leavings, a quick bit of googling on "firefox, greasemonkey and killfile" will soon set things right.
Behold:
Comment by jspreen blocked. [unkill]â[show comment]
I never thought I have to use it on a science blog. Political blogs, yes.
I always wondered why so many dogs find it appealing to eat each other's droppings. Maybe there was an evolutionary benefit to dogs who did so?
"Raphael, it just ends in frustration if you think in any way your obvious questions will persuade it to think scientifically."
Oh, I don't really think that I can convince people like jspreen to think scientifically- I just sometimes find it amusing, and sometimes even interesting, to watch their responses or lack thereof to reasonable questions.
Raphael, I'm saving the links to your comments here and here so 1) everyone can see what a weaselly little coward jspreen is AND 2) they can save themselves ten minutes of typing in reply to the little energy creature. He reacts to "show us your evidence" like bleach does to ammonia.
Could that be simply because there's not a single trace of science to be found in today's medicine?
But may I invite you to have a look at this and think of a smart scientific comment?
Should I find it ominous that they felt the need to specify "surviving patients?"
A small trial of fecal enemas for ulcerative colitis was also highly successful. All 6 UC patients who underwent fecal enemas from healthy donors went into long remissions (in fact it's tempting to say "cured", since UC relapsed in none of them over years).
May be some time before this hist the supermarket shelves - although the marketing possibilities of using celebrity 'donors' would be enormous:))
Orac on Hamer and New German Medicine
I stumbled upon that article yesterday. And jspreen is all over that one like stink on shit, too.
Yeah, I'm all over the place. Some love it, others don't. Well, such is life, I guess. Here, more heaps of fascinating information! You're welcome.
Hi All,
My 23 year old daughter was just diagnosed today with c diff. We live in a very rural area and don't have easy access to really good health care. She has been very sick now since the spring of 2004. Over the past 4 years she has had about 6-8 bouts a year that she would be very sick for 2-3 weeks,and then a few weeks of "FEELING GOOD". Each time she would be prescribed Flagyl and she responded very well for the 10 days and then even was feeling fair till her next relapse. This past year has gotten so bad that we finally headed to a bigger city and got into a gastro specialist. That took 6 months to even get into see them. After a 2 hour consulation, he ordered extensive tests be done. It was very frustrating as she had already had a colonoscopy done in 2005 which came back normal, stool testing that each time came back normal, blood work, etc. etc. By the time we got to see him, she had been bed ridden for over a month, diahrea with lots of blood and mucous, excrutiating lower left abdominal pain and nausea. I truly felt she was going to die on me and yet I felt helpless to find answers because after 3 1/2 years of having every blood test and other tests known to man we still had no answers. well the phone rang and the doctor confirmed she had c diff infection and only mild inflammation of the colon. He has started her on Flagyl for 10 days and she may have to go on another antibiotic after that. I have been searching all day on the web about this disease and it scares me to think of her not responding to the antibiotics and having a full recovery. If anyone out there has a story of similarity to mine I would sure like to hear from you. I would like to hear from others that have been diagnosed with this and their prognosis and progress of the disease. I know it is easy to make jokes about this and to keep my sanity I can find humor in them too, but I have to say if this type of treatment can help someone with this disease, then I hope trials for research are available to infected patients as it is a HORRIBLE DISEASE that needs to be studied so people are not mis diagnosed and suffer for years unnecassarily. Hope to hear from others on this disease. Thanks
Hi, Well to all that made fun at us with this miserable CDAD I say go ahead and have fun. To those in agony that I have had for 2 1/2 years, begging Doctors for relief, Flagyl did nothing, Vancomycin at the max dose of 2000 mg a day could not stop it for more than 12 hours before one could hit the eye of a needle at forty paces.
I had my fecal implant done on Feb 11 and have been off of all Vancomycin since, no more 20 to 30 trips to the bathroom! My bowel is slowly healing up and my pain is down 90% and continuing to improve. I have a normal bowel again all because I took a 7 day course of antibiotics. The thought of loosing the bowel is a lot more icky than this proceedure. Most people don't know this but the proceedure is painless, takes a couple of hrs, spend a nite in a hotel and you can go home. I have seen more Doctors and Hospital ER's than I care to see. Dr. Louie has a good track record and is truly a person who has a heart. There is no price for this proceedure that is high enough yet the medical system will not pay the Doctor doing it. The cost of Vancomycin was close to $3,000.00 for a 45 day supply. That is why no other Doctor will do it. Think about it. Have a good day, the passage out of the Bible says "it is better to Give than recieve"! In my case I was much happier to recieve it.
Hi, other than the obvious gross factor, why not fill an empty capsule with fecal matter from a healthy donor and swallow with a stiff chaser?
Should I find it ominous that they felt the need to specify "surviving patients?"
Maybe this is why dogs eat other dogs' shit from time to time.
A canine fecal transplant keeps the pack's stomachs in order.
David,
I hope you are still looking at these posts. Since I posted in January about my daughter, this year has been HELL! My daughter has been treated with vanco, xifaxin and still nothing has worked. It is now November 2nd 08 and we have found a doctor ready to perform the fecal transplant through a naso gastric tube. She has lost over 50 pounds and cannot eat or drink anything. Her gall bladder failed and also the sphincter of oddi, all of which needed surgery to correct. The scariest part is how many false negative c-diff tests came back, robbing us of valuable time. The transplant will take place on this Friday and I pray this will end it. I will be sure to let all know of the outcome. With all of the false negative tests for c diff, they even wanted to commit her to a mental institution!! After talking with this doctor who is performing the transplant, I believe this will be the miracle I and my family have been waiting for. Please let me know how you are doing to date and I will post results of her progress.
I've just read all the comments here and I'm putting up a real fight here in FL. My Dad got C-Diff at a huge hospital well known for their huge Trauma center...and now it's huge for C-Diff as well!! It should be renamed Drama Center...because everyone is crying from the damn C-Diff laying around to easily catch! Anyhow we too are going to try this as a last resort for my ailing Dad. Hopefully within the next week or two. Pray for us...as I pray for all infected!
Hi KLJ,
I will pray for your family as well as my daughter. It is a horrible disease and my daughter is heading to Duluth this week for the transplant. The doctor she is going to have do it is Dr. Timothy Rubin and his office number is 218-786-3985. I will post our results and wish the best to you. Dr. Rubin will be the guy to make it happen!!!!
KLJ,
Thought you might want to read this article. Don't let this go too long, this "BEAST" can kill you!!
What is a stool transplant?
Gastroenterologists at the Duluth Clinic offer a stool transplant procedure to people who have symptomatic recurrence and documented Clostridium difficile (C.diff) colon infections despite multiple attempts to treat them with traditional antibiotic therapy.
The treatment involves the infusion of a normal healthy stool from a donor that replenishes the normal flora of a patient's colon. We have performed more than 60 stool transplants with a high success rate.
Dr. Johannes Aas and Dr. Charles Gessert published the results of an
18-patient study in 2003.
For more information
Call our Gastroenterology Department at (218) 786-8555 or toll-free at (877) 266-5555. Office hours are 8 a.m. to 4:30 p.m. Monday through Friday. You can also email gastro@smdc.org.
Dr. Timothy A. Rubin, a gastroenterologist, currently performs the majority of the stool transplants at the Duluth Clinic.
To be a candidate, the minimal criteria are:
⢠At least two or more documented episodes of recurrent symptomatic C. diff diarrhea, despite adequate treatment; and
⢠Recurrences must have been treated appropriately with antibiotics. This includes an adequate trial of Flagyl (metronidazole) and vancomycin.
Here are some frequently asked questions:
1. What is a stool transplant?
A gastroenterologist uses a syringe and a small feeding tube to flush a small amount of prepared stool (30-40 cc) from a normal, healthy donor into the patient's stomach. The donated stool has been tested and prepared by our microbiology lab. The natural flora from the stool then repopulates the patient's colon.
2. How long does the outpatient procedure take?
You can expect to spend three to four hours at our offices. This includes lab tests, an X-ray and the procedure itself.
3. How long does it take to recover from the procedure?
There is no recovery time for this outpatient procedure. Patients walk in and walk out of the office. There are no restrictions on what you can eat or drink after the procedure.
4. How long does it take for the stool transplant to work?
Every patient is different. Some patients notice results within days while it may take several weeks in others. A follow-up lab test is usually done within four to six weeks.
5. How do you know that the donor stool is OK for transplant?
The donor's blood and stool are tested before the transplant date for C. diff toxins and other infections.
6. How is a patient referred?
You can make a self-referral or be referred by your primary care doctor, a gastroenterologist or an infectious disease doctor. You can call (218) 786-3801 or toll-free at (877) 266-8555.
7. Is a stool transplant covered by my insurance?
You need to check with your insurance company. Since most patients are traveling to Duluth, which is outside their health plan's network or tier, there usually is a fee. Our staff can help you navigate your insurance coverage and fees.
Hi Everyone,
Today is Thursday, 11/20/08 and my daughter just got home from Duluth after having the transplant by Dr. Rubin on Tuesday the 18th. She is doing wonderful!!!!!! She is eating any and everything, having normal bowel movements, nausea is down 90% and her energy level is through the roof. I still can't believe it after being sooooooo sick for sooooo long that this simple procedure has helped her this much in such a short amount of time. Our prayers have been answered!! She said the worst part of the whole procedure was placement of the naso tube but even that wasn't bad. I will post again in another week and recommend anyone with this hideous disease to consider making the trip to Dr. Rubin. My daughter said he was a very caring man and his nurses were wonderful. God Bless you all.
Hi Mamac,
I am happy to hear that your daughter is doing well. I recently just stopped taking Flagyl on November 17th, 2008. I am awaiting my test results from a new stool test. I have never been in so much pain with bowel movements until I was diagnosed with C-diff. I got it from possibly overuse of antibiotics or possibly a patient. I am healthcare worker and have been around numerous c-diff patients but never really understood how debilitating it can make you feel. I knew I had to get healthy bacteria in me immediately. My doctor did not diagnose me for a week and I thought I was going to die. I have been taking 15-20 probiotics along with eating yogurt, bananas, potatoes, rice, and bread as tolerated each day. My intestines did not like the probiotics and I vomitted. I made sure to take probiotics with rice between my flagyl 500mg/day x 3. Since, I have read these post I may have to try the implant. Thank You, for the information to contact the physician. I do not understand why the physicians do not tell you this is a possibility. I see elderly people die all the time with the preceding diagnoses of c-diff. Anyway, please update on how your daughter is doing. Thank You, so much!
Hi Mamac,
I am happy to hear that your daughter is doing well. I recently just stopped taking Flagyl on November 17th, 2008. I am awaiting my test results from a new stool test. I have never been in so much pain with bowel movements until I was diagnosed with C-diff. I got it from possibly overuse of antibiotics or possibly a patient. I am healthcare worker and have been around numerous c-diff patients but never really understood how debilitating it can make you feel. I knew I had to get healthy bacteria in me immediately. My doctor did not diagnose me for a week and I thought I was going to die. I have been taking 15-20 probiotics along with eating yogurt, bananas, potatoes, rice, and bread as tolerated each day. My intestines did not like the probiotics and I vomitted. I made sure to take probiotics with rice between my flagyl 500mg/day x 3. Since, I have read these post I may have to try the implant. Thank You, for the information to contact the physician. I do not understand why the physicians do not tell you this is a possibility. I see elderly people die all the time with the preceding diagnoses of c-diff. Anyway, please update on how your daughter is doing. Thank You, so much!
Hi Brenda,
So sorry to hear you are not feeling well. Dr. Rubin feels as though my daughter had recieved so many anti-biotics by IV that it killed off all of her good flora and bacteria. She too had tried many probiotics to no avail. Dr. Rubin said that there are so many bacteria and flora in the GI tract, that to this day have not been identified nor do we know what role each of these play in our GI system, that the only real way to replace them all is to use the stool of a healthy and SCREENED donor. I do not know why physicians don't let people know of this option, but the word needs to get out! The entire procedure costs less than $1000 including the donor's screening costs, heck, it costs that for a 1/2 round of vanco. It makes me sad and angry to think that this could all be about money but seeing the amazing results truly makes me wonder. It is horrible to think of others out there suffering needlessly and many of them dying or having their colons removed before giving them this option. You will be in my thoughts and I will keep you posted on my daughter's progress. Today for the first time in over a year, my daughter and I are going out for lunch together! I am loking so forward to it. Take care of yourself and let us all know how you are doing.
I found this article today and thought some of you would like to see it. Very scarey!!
September 17, 2008 - Pittsburgh: C. diff's ground zero (The Hamilton Spectator)
Read the article on The Hamilton Spectator site.
Naomi Powell
Over and over, patients were wheeled into the operating room of the University of Pittsburgh, Presbyterian hospital, their colons grossly inflamed.
Surgeons cut the infected organs out, often to no avail. Most patients died shortly after they were wheeled out.
Dr. Ramsey Dallal, then a young surgical trainee, started digging through the hospital records. Sixteen colons removed in 2000, all riddled with the same bacteria, Clostridium difficile.
C. diff? For decades, the bug had been in every hospital, the pest behind painful diarrhea and sometimes worse. But not normally a serial killer -- until now.
C. diff had morphed into a deadly new strain, one resistant to antibiotics, targeting the elderly and capable of wreaking havoc in hospitals.
An old bug, with a frightening new identity.
"You're seeing these patients come up over and over again, people dying of C. difficile colitis," said Dallal, now a bariatric surgeon at Philadelphia's Albert Einstein Healthcare Network.
"It was strange ... You've never been taught that this is C. diff."
The 2000 Pittsburgh outbreak was the epidemic strain's first known appearance in North America. It wouldn't be its last.
The superbug has become the most widespread C. diff strain, causing outbreaks in hospitals across the United States, Canada and Europe.
Each time, it has left doctors puzzled by how it passes through some patients quietly, without causing any damage, and rips through others, killing them within days and sometimes hours of its appearance.
At the peak of the Pittsburgh outbreak, the number of C. diff cases in the hospital tripled. The number of patients facing life-threatening symptoms doubled.
"Our patient-safety people were freaked out by the fact that we were having deaths related to C. diff," said Dr. Carlene Muto, medical director of infection control at the hospital. "We believed this to be a nuisance disease before this happened."
Muto expected studies on the Pittsburgh outbreak to send shock waves through the medical community. But though it would forever change Pittsburgh's understanding of C. difficile, the outbreak caused barely a ripple outside the city.
"The interesting thing is for quite a while, no one believed there was anything new or novel with C. difficile disease," Muto said. "It took us a long time to get our case control study paper published because nobody was that interested."
Pittsburgh's story grabbed the attention of at least one expert.
When Dr. Dale Gerding heard about the 16 C. diff colectomies in the hospital, he "just about fell over."
"I couldn't believe it. I had probably seen two or three patients in 20 years undergo a colectomy for C. diff and they had 16 in one year?"
A professor of medicine at Chicago's Loyola University, Gerding had collected C. diff samples since the early 1980s, when a test was first developed for the bug.
The strain that attacked in Pittsburgh -- called NAP1, BI or 027, depending on the system used to identify it -- matched a sample Gerding had collected in 1984. Both produced a pair of toxins (labelled A and B) and both had a genetic mutation that prevented a "repressor gene" from limiting the amount of toxins produced.
There was one crucial difference: The Pittsburgh strain had acquired a stubborn resistance to a class of antibiotics known as fluoroquinolones.
Among the most common hospital antibiotics, fluoroquinolones came into broad use in the 1990s as a treatment for respiratory illness.
NAP1's new armour against them made it enormously powerful. While the fluoroquinolones swept aside protective bacteria in the gut, NAP1's resistance allowed it to stay and thrive.
It could grow and release its toxins -- the bug was later found to produce 16 times more toxin A and 23 times more toxin B than other strains. It could inflame the bowel and eventually pass on more spores through diarrhea, further contaminating the hospital environment.
Outside the body -- where the bacteria struggles to survive in oxygen -- the spores provide an ideal shelter. They allow the bacteria to hide, dormant, inside a tiny indestructible vessel that can withstand most hospital-grade cleaners.
The bug only comes to life as active bacteria inside the colon, where it is believed to feed on the carbohydrates in our bodies. It grows rapidly as long as it has enough to eat, then hits a stationary phase, when it releases toxins and more spores.
Exactly how and when the bug evolved to outsmart fluoroquinolones remains a mystery that experts chalk up to the constant, spontaneous genetic evolution of an ancient organism.
"It's got different armour," said John Bartlett, an expert on infectious disease at Baltimore's Johns Hopkins University.
"It causes disease differently and now it's an epidemic in the U.S. and in Europe and all over the place.
"A roaring epidemic. But it's an old bug. That's what's interesting."
If Pittsburgh was a wake-up call to C. diff's heightened potential to kill, Quebec was a screaming alarm bell. The bug's deadly attack on that province, which began in 2002, would eventually kill at least 2,000 people.
For Dr. Mark Miller, it began in April of that year, with a scenario eerily similar to Dallal's in Pittsburgh.
"One of the colorectal surgeons called me and said, 'I just took out my second colon for C. diff in a month,' " said the director of infection prevention and control at Montreal's Jewish General Hospital. "He asked me if there was something going on."
Miller asked around. His colleagues at other Montreal hospitals were all experiencing the same thing: higher infection rates and deaths.
That fall, the doctors called an emergency meeting. Over pizza, they mapped out an ad hoc system to track the number of patients dying of C. difficile.
"We met again in December and put our numbers down on the table," Miller said. "We were absolutely horrified."
At the height of the Quebec epidemic, 15 per cent of all patients infected with C. diff died. In Miller's hospital alone, there was an average of 40 cases of the disease and seven deaths every month.
And everywhere in the region, doctors were finding that traditional outbreak remedies such as handwashing and isolating patients weren't enough, according to Dr. Michael Libman, director of the infectious disease division at McGill University Health Centre.
"The total failure of our initial infection control measures was a shock," he said. "Essentially, our getting together as a group was a bit of a panic reaction. We were getting together because we didn't know what to do."
Controlling the new strain and removing its spores from the environment required a previously unseen level of vigilance.
Visitors and staff draped in gowns and gloves became a common sight in the hallways of Montreal hospitals. A new set of rules governing handwashing and cleaning techniques was enforced. And the Quebec government sent teams of professionals into hospitals to ensure proper cleaning, isolation and antibiotic management.
"What we've learned in Quebec is this bug is extremely unforgiving," Miller said. "You make one or two slip-ups in infection control and it comes back with a vengeance. It just doesn't stay put."
Around the same time that hospitals were fighting NAP1 in Quebec, the superbug was launching attacks in Georgia, Illinois, Maine, New Jersey, Oregon and Pennsylvania. Though some of the bugs had slightly different DNA fingerprints -- indeed, Pittsburgh's bug bore a slightly different DNA pattern than the one in Quebec -- all were versions of NAP1.
The damage in Quebec was severe enough to earn the bug a new nickname: the Quebec strain.
C. diff has since made its mark in Ontario, attacking first at the Peterborough Regional Health Centre in 2002. Though researchers never tested for NAP1 in the outbreak, in which 17 patients died either directly or indirectly because of C. diff, the strain was later found at the hospital.
The bug's more widely publicized assaults in Ontario came later. Burlington's Joseph Brant Memorial Hospital remains the site of the deadliest Ontario attack on record. There, 91 patients infected with C. difficile died in a 20-month outbreak that started in May 2006.
Other episodes occurred at the Sault Area Hospital in Sault Ste. Marie and Barrie's Royal Victoria Hospital in 2006, followed by Oakville-Trafalgar Memorial Hospital, Mississauga's Trillium Health Centre and Simcoe's Norfolk General Hospital in 2007.
These are only the incidents that the public knows about.
A tally by The Spectator shows at least 460 patients infected with C. diff have died at just 22 of Ontario's 157 hospitals since 2006. The newspaper is the only reporting source to date for Ontario C. diff fatalities.
Fighting the bug remains a struggle. Some say stricter cleaning regimes and updated hospital designs are necessary to minimize the number of C. diff spores. Others believe the best way to combat the bug is in hospital pharmacies, through the restriction of fluoroquinolones and other high-risk antibiotics.
Even then, it may be only a matter of time before the bug learns to outsmart the next drug thrown in its way.
"The expression some people have used is squeezing the balloon," Libman said. "You can squeeze off quinolone use, but the reality is some other antibiotic is going to have to pick up the slack. And then most likely the bug will just evolve to develop resistance to that one."
Few believe C. diff infection or death rates have peaked in Ontario, where the extent of the bug's presence will remain unknown until mandatory reporting begins on Sept. 26.
"It's not like the war against bacteria and infections is something we're going to win," Libman said. "It's going to go on forever."
As a 61 year old RN I can tell you the procedure was done in at least one university hospital in the late 70's with some frequency and with good results. It involved a small amount of stool from a healthy donor given with a milk shake to patients who had had their gut flora wiped out by multiple antibiotic courses. The patients were not informed of the "treatment". Ethics at that time were peer reviewed on the spot by the people involved in the patent's care.
As a 61 year old RN I can tell you the procedure was done in at least one university hospital in the late 70's with some frequency and with good results. It involved a small amount of stool from a healthy donor given with a milk shake to patients who had had their gut flora wiped out by multiple antibiotic courses. The patients were not informed of the "treatment". Ethics at that time were peer reviewed on the spot by the people involved in the patent's care.
Hi mamac and Paul,
I recently received a negative dx for c-diff although I continue to have symptoms. My right side(front and back) hurt really bad. I have a general physican and specialist seeing me for this diagnoses. My physician did not want to prescribe anymore antibiotics but the specialist did. The specialist stated that it can read negative but I can still have this bacteria. I also requested someone in the Kansas City, Mo, area who does fecal transplant. He gave the nurse a name of an infectious physician. So, I am trying to get in to see him asap before this continues to damage my organs. I have not started the second dosage of the medicine yet. The flagyl is just about as bad or worse then the infection it causes dizziness, cramping, vomitting ect..... I dread taking this again. I continue to eat only yogurt with 8 live cultures from health food grocery store and about 10 to 15 probiotics plus rice and bananas. Let me know how your daughter is doing. I am glad to hear that she is doing better. Brenda
Hi Brenda,
My daughter had many negative tests even though she still had the "BEAST" in her body. Have they put you on Vanco? If the flagyl did not work for you then Vanco would be the next med to try. When vanco failed for my daughter after 4 rounds of it, they then tried her on the combination of vanco and xifaxin. That combination seemed to work while she was on it but within 3 days of finishing it she was right back to square one.
It has now been 5 days since the transplant and she is on NO MEDS at all and doing great. Eating anything she wants, normal bowel movements and NO PAIN at all. It is truly a miracle. How long have you been sick for? My daughter was first diagnosed last January. Our entire family is still in shock at the improvement she has had after being so sick for so long. I know what you mean about the meds being terrible to take as my daughter fought to tolerate the meds each time, but I believe a lot of it was because they just were not working to start with.
I hope you find the help you need and my heart goes out to anyone with this horrible disease, but don't give up, there is a cure if the all the meds fail! I will continue to post her progress and I hope you will do the same. Take care of yourself and be strong.
Hi Mamac,
I went to my general physician today, November 24th, 2008. He said not to take the flagyl because my symptoms were mild. Unfortunately, the symptoms seem to be getting worse again. I did talk with him about the fecal transplant and he stated it was not a cure all and I could still get the c-diff. I told him if he was in this much pain that he would want to do it. I think it is a waste of time to take drugs if the fecal transplant works. I have had this since October 27th, 2008 and was on flagyl for 14 days. I was told in order to get the transplant you have to meet certain criteria? I am concerned about having this because it is affecting my life in many ways. I am still unsure what I need to do to protect my family friends from being infected with this bacteria except to tell them to wash their hands. Can you tell me what the infectious physicians have shared with you? Well, I am happy to hear your daughter is doing well. Will you be going for a follow-up visit to this physician?
Hi Brenda,
Unbelievable!! Don't take the meds because your symptoms are mild?? Allowed to flourish, c-diff will only get worse. You need to find a gastro doc that will put you on Vanco next. It is very expensive for a 7 week course but the good Lord willing it may work for you. If not, then you need to contact them out in Duluth. They do have a criteria for you to meet. On their website, they state they want to have at least 2 positive c-diff tests and be sure that the proper med regiment has been followed before considering a transplant. As far as protecting your family be sure to use lots of clorox for cleaning, stress hand washing with hot water and soap. Alcohol based cleaners do nothing to kill the spores. Transmission of c-diff has to be through the mouth, so stress to family members and you too to keep your hands away from your mouth. C-diff in the vegetative state will be killed in the stomach by gastric acids but the spores can survive and in turn invade the digestive system. Again, it is very possible that vanco will cure you since it has only been such a short time you have had this. Insist on continuous testing as a false negative test for the disease is very possible. If the symptoms are continuing and getting worse, you must be vigilant on requesting testing as my daughter had many negative tests but she was continuing to get worse daily.
Keep in touch and stay strong. You are in my prayers.
Hi Mamac,
Well, it is Sunday,November, 30th, 2008 and I am feeling better. I went to see the infectious disease physician and he prescribed a new medicine xifaxan 200 mg x 3/day for 3 days only. He said this medicine will stay only in the intestines and not enter the bloodstream. I finished my last dosage yesterday evening. He also told me to eat pasta. So, I decided to try the pasta. I have been eating primarily bananas, rice, bread, and yogurt. The bananas seem to make it worse for myself though. I still have some minor symptoms of burping and loose stool 2x yesterday. I will seem this physician in two weeks. He also recommended a specific probiotic that used to be a prescription but now is over the counter flora something. I had to special order this product at CVS pharmacy. Thanks for listening and your prayers. How is your daughter doing now?
Hi Brenda,
Glad you are feeling better and I hope it stays that way for you. I am very familiar with xifaxin as it was used on my daughter 2X's with the vancomycin for a 2 week regiment each time. She did great on it but within 3-5 days of completing the vanco and xifaxin, she would become very ill again. The vanco taken orally also stays in the digestive tract and not absorbed in the bloodstream.
My daughter is doing really well and we all enjoyed having her at the Thanksgiving table with us. Tomorrow it will be 2 weeks since the implant and she has had to take no meds. It truly is a miracle after being so sick and bed ridden for almost a year. I hope you are one of the lucky ones that is cured by meds and probiotics. My daughter was on the probiotic "ALIGN" but after a month there was no change. Dr. Rubin believes that my daughter just had no good bacteria left in her GI tract and the only way to replace it was with the fecal transplant. There was a study of 6 patients with ulcerative colitis that were treated with transplants done by enema which also looks promising but in this study they call it HPI, human probiotic infusion, which does sound nicer than a fecal transplant. Well, I am very happy to hear you are feeling better and wish you all the best. Be sure to keep me posted on your progress and I will do the same for my daughter.
Hi Mamac,
Well, I took my last pill on last Sunday. I have been doing pretty good. My stomache is a little sore still. I go back to the infectious physician within a week. I still have a few episode but not like previously. I am glad your daughter is doing well. It sounds like you suffered to long with this c-diff. How old is your daughter?
Hi Brenda,
Glad to hear you are still improving, that is so good. My daughter just turned 24 and she had her first positive c-diff test back in January '08, but she was sick for a couple of years befor that, she was just never tested for c-diff. They always kept saying she had irritable bowel syndrome. They tested her for Crohns, ulcerative colitis, ulcers, etc. etc. It has been a nightmare. At one point they even told her she should seek psychiatric help because nothing was wrong with her. I would ask if c-diff should be checked again and they would run a single test which sometimes would come back negative. I later found out that 3 tests, each one on a different day should be done. Even after having a positive test for c-diff, they would give her antibiotics that just made the c-diff worse. Anything that I ended up learning about c-diff was not from doctors but from my own research. It is so scarey how nonchalant some doctors are about c-diff, and unfortunatly I live in a very rural area and finding a good c-diff educated doctor was very difficult if not impossible. One time while my daughter was hospitalized, there were 12 people on her floor that had c-diff. I heard the floor doctor talking to the patient across the hall and he told this poor old woman that c-diff is a common infection and really nothing to worry about, just take the flagyl and you'll get better. I often think of her and hope she was one of the lucky ones that flagyl worked for. Well, we will keep in touch and I hope you and my daughter continue to get better and better each day. My daughter goes for a follow up visit with the gastro doc on Dec. 18th so I will keep you posted on her. Stay Well!!
Hi Mamac,
I'm very interested in your daughter's story sounds allot like my own. Did she undergo the spichter of oddi and gallbladder surgery prior to the transplant or is it to occur afterwards?
Hi Joey,
Yes, she had both prior to the transplant. Having those procedures only added to the c-diff problem as each procedure required IV antibiotics which in turn made the c-diff flourish. It is such a miserable cycle, but the antibiotics had to be used for the procedures. It has been almost 8 weeks since the transplant and yesterday she got the results of 3 consecutive c-diff tests and all were negative. It is so unbelievable!! Keep in touch and let me know if I can answer any other questions for you. Wishing you better health in the New Year.
Hi everyone,
I've been reading all that I can about fecal transfusions for the last year or so and only recently came across this website. It's nice to see that people are still interested in this subject, as I believe it to be a cure for many -- if not all -- bacteria-associated intestinal problems. I myself have IBS-like symptoms (hard stools, bloating, inflammation) and am looking to possibly do a home version of fecal bacteriotherapy in the near future.
I was intrigued to read one of the posts that described an alternative way of doing the procedure:
"Apparently the "healthy stool" was introduced by mouth, mixed in with chocolate milk! (I will let that thought linger without comment.)"
I'm wondering whether or not this might be a preferable method to restoring bowel flora, as the donor's flora would reach the entire digestive tract. That is, barring any acid-bath deaths in the stomach, of course. Perhaps the milk somehow prevents this from happening. Anyway, I wonder what the risk would be in consuming a healthy, disease-free donor's stool orally? Thoughts anyone?
BTW, any and all information from individual experiences with this therapy is greatly appreciated.
Hi rk900,
My daughter had the transplant done out at the Duluth Clinic back on Nov. 18th. Her husband was screened for many diseases and was the donor. They mix and blend the stool and filter it. While that is being processed, they placed a naso gastric tube into her stomach. They then took an xray to be sure the tube was correctly placed. They then injected about 4 TBSP of the stool, removed the tube and it was done in less than 2 minutes. Th doc out there believes that when someone was as sick as she was, then treating the entire GI tract is imperative. In order to reduce stomach acid that could kill the good bacteria, my daughter had to take a prilosec the night before the procedure and then another one the morning of the procedure. She said the hardest part of the whole procedure was having the tube put in, but there was no "ICK" factor for her at all. I only wish people could have actually known and seen how very sick my daughter was for over 18 months. She had been treated so many times for it and yet nothing worked. Her last resort was to have the colon removed. I am just so thankful that this procedure was available for her and I would recommend it to all that have this Beast within them. Let us know how you make out.
Thanks for your story, mamac. I'm glad that the fecal therapy works for people in such dire straits as your daughter was. I truly believe it is the answer for people with any kind of GI disorder that may be bacterial, even if their situation isn't life-threatening. An answer for IBS, chrohn's, ulcerative colitis, and gut bacterial infections of all all kinds could possibly be found in the fecal transplant -- if only our medical community showed more interest in it!
In my case, I have "mild" symptoms -- hard stools, gut inflammation, bloating, and trapped gas. My stool test (taken several months ago) revealed that the parasite "blastocystis hominis" was found in great numbers. This is believed to be the cause of my symptoms by my doctor. I think it may be, as well.
After a few courses of flagyl, I'm not interested in taking any more antibiotics. So now I am turning to what I believe to be the ultimate cure -- the fecal transplant. All of the experiences with this procedure that I've read about online have shown very good results for ulcerative colitis, crohn's, c-difficle, constipation, and the like. So I'm certainly hopeful that this is an answer for my gut problem.
I have a friend who I've selected as a donor based on the fact that he's never -- I repeat NEVER -- had any sort of gut/digestive issue in his 28 years of living. Lucky dude. And I'm hoping his "luck" will pass on to me. :-0
I have read about these in the past and I have an aging father in Bradenton Florida that also got C-diff from a local hospital stay and a doctor that kept him on antibiotics too long to be able to resist it. He has been on Vancomycin now for almost 2 years...... yes i said 2 years. I have been looking for someone here in florida to do this type of transplant since i read about it 2 years ago.... who has a referral????
What great things can be done for something so simple....too bad our culture is so caught up in the yuk factor...especially our medical people. It is great to hear how you all have persisted and the good outcomes that are realized!
Hi Nancy,
My daughter and I live in Maine,and we had to make the trip to Duluth as it is the closest place we could get it done. I contacted so many facilities in N.Y., Mass.(Boston General),Duke University in N.C., etc. etc. No place did or would do the transplant. I was blown away that not one of the large medical centers offered the treatment. Not only did they not offer it, I felt that they didn't even want to talk to me about it. My daughter is the first patient from Maine to have the procedure done. I hope you can find someone who is familiar with the procedure close to you but I highly doubt it. I would recommend Duluth no matter where you live. Getting there is an expense but one that was worth it for us. Good luck to you and keep us all posted.
Just wanted to let any frequent readers of this comment section that I'm currently consulting a doctor in Salt Lake City about the fecal transplants. Mainly, he's giving me advice on which tests to perform on my donor. If my donor checks out, I plan on doing the procedure at home.
Again, my symptoms are IBS-C, and I'm approaching this whole procedure with the idea that if I acquire stool from a healthy donor that's regular and has no GI issues, his flora will implant in my gut and make me well again. A healthy gut with no bloating, trapped gas, inflammation, and hard stools would be a dream come true for me.
Wish me luck!
rk900,
Best of Luck to you!! My sister who has UC is considering doing a tranfusion at home so be sure to keep us posted.
Oh joy! Lets all sign up. This sounds so clinically inventive, especially that nasogastric tube. How "advanced."
Just ignore the probiotics and preventative health info that has been available in massive sums of data in every culture, and prop up those ivy league grant proposals. What could be more profitable than loads of antibiotics and fecal transplants by nose tube?
I was wondering exactly how you were planning to do this at home as I have had colitis for 20 years and would also consider it. My lovely wife of 30 years of marriage would be my donor. PS. I am sure we have swapped much bacteria over the years.
Erin,
Ignore probiotics, preventative health info?? Probiotics tried for over 6 months and as far as preventative health info, my daughter aquired c-diff while in the hospital and had never heard of it previous to catching it in the hospital. All I can tell you is after $20K of meds and all the preventative health info, the only thing that worked for my daughter was a fecal transplant by naso gastric tube.(which by the way only costs around $300). I pray that you or any of your family members are never infected with this disease, but if you do and cannot rid yourself of it by conventional medical treatments, would you let them remove your colon without trying this treatment???
mamac,
I appreciate your detailed description of the fecal transplant. Did your daughter take a course of antibiotics as a prep before the procedure?
Brad,
Yes she did. Even though she had been on numerous antibiotic regiments that had failed repeatedly for over a year, the doctor in Duluth wanted her on a 7 day course of Vanco to keep the c-diff somewhat under control until she could get out there. Even on the Vanco for 7 days prior, she said it was a very, very long plane ride out to Duluth. The Vanco was stopped the night before the procedure.
mamac
For those interested in a home infusion protocol, here is a link:
http://hpit.googlegroups.com/web/2005%20home%20infusion%20protocol.doc?…
I have still not gone through with the procedure due to complications with my donor and his availability. He returns in late March and hopefully he'll be able to get the blood/stool tests done so we can move forward.
To those (like Erin) who think this is some sort of "advanced" medical procedure, with a little research you'll find that it's not. In fact, the idea is simple: get healthy stool into an unhealthy GI tract. The cost for that -- depending on your constition -- could be very minimal (even free if you have a healthy partner). All that's needed is healthy stool ($0.00), donor tests ($10.00 sliding scale fee for my donor) and prilosec ($12.00), the latter of which may be uneccessary. Mix with milk and down the hatch! No nasogastric tube needed, and it's basically the same effect. Might gross most people out, though.
Alternatively, enema supplies aren't very costly at all.
Fecal bacteriotherapy is not some wiz-bang, top-of-the-line, get-you-in-debt-fast procedure. It's the complete opposite and may be the quickest, most effective fix for all sorts of digestive diseases!
I'm female, I'm only 25 (it's not reserved just for the elderly and weakened hospital patients!) and have been battling C. diff recurrences off and on (mainly on) since July of 2007. It happened because I got MRSA on my right leg, and the antibiotics for that absolutely wiped out my "healthy" flora.
Flagyl failed repeatedly. Eventually an infectious disease specialist literally saved me with a long course of Vanco, that worked well, and I went relapse free for awhile. Until...yep, the diff came back :( Back on the Vancomycin...which works, then within a week of stopping, I relapse.
I finally "qualify" for this treatment. It makes me pretty squeamish and I'm a little scared, I don't know why, but it's my only option left now. And I'm relieved to be getting it, at last. This illness has ruined two jobs for me, and I'm trying to start my career but I keep stumbling into relapses! Thanks for this article Tara.
Hi Anonymous_sicky,
My heart goes out to you. You are the same age as my daughter that I write about above. I wish you all the luck in the world with the fecal transplant. My daughter was also squeamish about it but she said the worse part of the procedure was having the naso-gastric tube put in. The actual infusion was painless and nothing nasty about it at all. Be strong and if this works for you, you will have your life back. God Bless you and keep us posted.
MamaC,
Thanks for your kind words. I read your daughter's story above. What she went through is one of the worst stories I've ever heard, I'm so sorry. How is she now?
The naso-gastric tube is the part that scares me most. I'll have to be sedated :P
Hi anonymous_sicky,
She is doing good and again, just got 3 negative tests back so we sure hope it is over for good. It really has been a nightmare for her. I hope it ends for you soon. Are you scheduled for a transplant yet? Don't be scared of the naso gastric tube, it will be well worth it but if you ask to be sedated they probably will. Please keep me up to date on your progress and I will pray for your complete and speedy recovery. Has the c-diff caused you debilitating nausea? Besides the debilitating diahrea, she suffered with debilitating nausea and abdominal pain. Best wishes to you.
Does anyone know of a doctor who will perform this procedure in south Florida? I'm trying to save my mother who is 89 and was otherwise quite healthy before C-diff struck. Many thanks.
I was just re-reading some of the blogs and saw the entry about using only 4 tablespoons of feces for the nasagastric tube procedure. I wonder if feces paste could be placed in gel capsules to equal 4 tablespoons. Swallowing gel capsules sounds a bit easier than either the gastric tube or the feces milkshake. I doubt whether my 89 year old mother could survive a trip to Duluth...
I'm also wondering if researchers could explore using a blue light diode in their colonoscopy tubes. The blue light wavelength is being used to kill MRSA in superficial wounds. Maybe it would also kill c-diff spores. But that wouldn't help c-diff in the small intestine, unless you could also swallow a blue light diode. I'm having trouble shutting-off my brain tonite - this has been such a nightmare.
Hi Kay,
The capsules route (very inventive, BTW) sounds more complicated than the milkshake route, IMO. I myself am going for the milkshake when my donor gets home and gets tested (only a week away!). I don't have clostridium, only IBS.
As to the blue light diode stuff, I have no knowledge of that, but it sounds like something that certainly warrants some research.
Good luck, and keep us updated!
Hello rk900,
I'm excited for you!! Please tell us how it tasted and whether it caused you to gag/vomit, and just how much you put in the milkshake. If it were me, I'd probably wait until I was very thirsty before drinking, and maybe add some of Trader Joe's strawberry flavored kefir to the mix, and maybe even a dash of ginger ale. I wonder if a series of daily shakes with small amounts of poop would work. In any event, it can't possibly taste any worse than colonoscopy preps. I have no doubt that it will work for you. Thanks for your post.
Hi,
My Dr has recommended this to me (Centre for Digesttive Diseases in Sydney). I am not suffering from the same issue as the patients in the study, he is recommending it for Blastocystis hominus. However I cannot find any litreature on this type of treatment being used on my condition. Can you point me to some? Also where can I find some emperical data on this treatment, and documentation of the risks involved?
Thanks
Marie
Kay,
I'll let you know how it goes! If one milkshake doesn't do the trick, I'm prepared to try more -- or go the enema route if needed. I'll certainly try to make the shake taste as good as possible!
Marie,
That's really interesting! I'm doing the procedure for the SAME parasite (blastocystis hominis). As far as I know, there isn't any literature on fecal transplants for blasto, but there is plenty on doing the procedure for clostridium, ulcerative colitis, crohn's, and constipation -- more and more research suggests that these conditions are due to gut flora imbalance. I think a blasto infection can also be lumped in the same category. The parasite affects us because our gut flora isn't balanced enough to keep it in check. Check out Wikipedia for more info on fecal transplants along with some good links:
http://en.wikipedia.org/wiki/Fecal_bacteriotherapy
There's a good PDF file on that page of a study done by Borody and others. Scroll down to references and click on the PDF for "Toying with Human Motions."
As for risks, I've never heard of one case of ill health following a fecal transplant -- only better health. Seems to be a safe procedure as long as the donor is healthy without any diseases.
BTW, my donor is home, and he's getting tested tomorrow. After he returns in a week, we'll have the results, and I can finally go through with the procedure!
Oops, I should probably clarify my situation: I have symptoms of IBS -- hard stools, bloating, trapped gas -- that my doctor believes are caused by blastocystis hominis.
UPDATE: My donor's tests cleared, and I just implanted my first fecal infusion via a fleet enema (rather than the doing the milkshake). I used a thumb-sized portion of stool, shook it up in a jar with a little water, and then strained this concotion into a pourable container. It resulted it about 1/8 of a cup of stool liquid. I then added a little more water to make it runny, poured into an empty fleet enema, and squeezed it in!
I decided to experiment with this method first, rather than going the milkshake route, because I wanted to test its effectiveness. Tomorrow, if I feel no difference, I'll acquire more stool from my donor and try the milkshake (which, at this point, sounds easier and more appealing than the enema).
Right now, I feel fine. No real difference. I'll update as things (hopefully) progress!
It's the morning after the enema implant, and I already feel more calmness in my large intestine. I'd say, overall, I feel 50% better -- in less than 24 hours! I don't want to jump to conclusions, though ... I just hope things continue to improve.
One thing I noticed with the fleet enema method is that the implant was only able to reach the first bend -- maybe a little further -- of the large intestine. So, while the desceding colon and horizontal part of the large intestine feel very calm without bloating (awesome!), the ileocecal valve and ascending colon is still bloating and trapping gas.
So it seems that I either need to clear the colon of all feces and implant a deeper enema (which may not reach the ileocecal valve). OR I need to try the milkshake to treat the entire GI tract. I'm opting for the latter choice this morning once I acquire the "donation."
All things considered, it's amazing how quickly things have improved. Last night I woke up refreshed and felt absolutely NO abdominal tension or bloating. I can't even remember the last time I felt that way. Soon after that (about an hour later), I began to experience bloating and trapped gas around the ileocecal valve and ascending colon. As I said, the symptoms were isolated in those areas, while the rest of my intestine remained symptom-free!
Hi MamaC
Realizing now that I never did post back. I got my fecal transplant done, via enema instead of the naso-gastric tube, on April 7th. That's just how this doctor does them. It seemed to work great at first, and I was feeling absolutely fantastic until this morning. It looks like I'm definitely relapsing again so the doctor says we'll be doing another transplant this coming Thursday. Hopefully this time it will stick!
To answer your question, I do get quite a bit of nausea with the C. Diff. It's actually the worst in the days leading up to relapse, and then it tends to get slightly better once the diarrhea starts. But it isn't until I go back on Vanco that the nausea returns.
I hope your daughter is still doing well!
hello rk 900
i have been with severe diarrhea for over 10 years now. for the last 2 weeks have done lots of research about fecal enema . thank you for posting all your experience since i am planning to do it at home also .
your last post is from april11, can you tell me how you are doing ? did you only do one fecal enema ? Did you lie down for a while ? or had the urge right away of going to the toilet ? you did this at night before going to sleep ?
my donor is my 16 year old daughter, she is very healthy and has never taken antibiotics . did you use the fleet water ? or just regular water ?
thank you and i really hope to hear from you
UPDATE
So I haven't posted in a long while re: my improvement with the fecal implant. I would have posted a long time ago if I experienced any more improvement than the initial implant. Unfortunately this has not been the case, and I still experience the same symptoms as before depending on what I eat (more on that later). I even tried a few shit milkshakes (!) and no notable improvement resulted. The most intestesting aspect of this latter experiment was the similarity in smell of my poo compared with my donor's the day after the experiment. I did have a nice bowel movement or two, but reverted back to hard stools soon thereafter. And the bloating hasn't ceased at all so far.
The confounding factors that could have affected the outcome of my home experiments are as follows:
1) The enema I performed did not treat the entirety of the large intestine. It was a small amount of liquid (plain water mixed with strained stool), as opposed to the quart or so that seems to be the norm for deep enemas.
2) I didn't do a colonic "lavage" beforehand, so I think the flora only reached the first bend of the large intestine -- maybe the implant was blocked by stool already hanging out there.
3) I didn't do any sort of antibiotic regimen before the procedure, so whatever flora I implanted was competing with the flora already thriving in my intestines.
4) I didn't take prilosec before downing the shitshake, which could have resulted in a stomach-acid bath death for much of the ingested flora. Prilosec is definitely always a part of the nasogastric tube procedures that I've researched.
It's interesting that I initially felt very good from the first implant via enema, but like "anonymous_sicky" above, I soon reverted back to my old symptoms. Maybe I need to perform more enemas/milkshakes to solidify the flora replacement. Or maybe I need to reevaluate the root of my symptoms, which may not be related to gut flora as much as I think they are. I seem to have the most problems with high fat meals, so maybe I need to address fat digestion problems.
For "kd" above: I didn't use the fleet water, as this is usually some sort of harsh laxative that would make it hard to retain the stool. I used a very small amount of water (maybe 2-3 ounces) mixed with a concentrated "stool water" that was made by straining the stool mixed with a little water (as described in an above post). I had no problem retaining this.
Good luck!
I'm also curious how "sicky" is doing since doing another implant. How are things, sicky?
Hi y'all...got stung by a fireant in Florida late March 2009...foot infection ensued within 5 days...saw a doc in Tenn. then P.O clindamycin 300mg x10 days(i only took 7 days)..then the next day, the foot was still badly infected...then had IV antibiotics-one dose...foot healed up, then 3 weeks of watery stools turning to loose to semi formed diring the last 3-4 weeks...last three stool tests..first one neg, then last two pos for C-diff... my symptoms are very mild at this point but still getting loose to semi formed stools daily x 3/day...another doc gave me a script for Flagyl which I am delaying filling...I am thinking of trying colloidal silver, Nascent iodine and a certain garlic pill or garlic cloves to fight the c-diff...alternatively I am just now reseaching this blog and this sounds like the way to go...with the labs results which I had printed(I used to work as an RN for 9 years)there was a flag for a clinical trial drug called par101..I called the research nurse today and its a double blind with Vancomycin and the PAr101 new drug...she states the par101 is 100% effective against the c-diff...i am debating which way to go...I realize that I don't want to feck around with the c-diff but i also don't want to get caught in the cycle of antibiotics and recurrence...the stool transplant sounds ingenious and just good common sense...i will be following this blog in the future....Govindadaz
This procedure is not available in my area, but I wish it was. I'm a Case Manager at a local hospital and one thing I handle is discharge planning for a patient. The current treatment is oral Vancomycin, Oral Flagyl, and some oral probiotics. Do you know how much a few weeks of Oral Vancomycin cost? Try 1,200 to 1,600 dollar. And Medicare or most private insurances will not pay for the oral form of Vancomycin as outpatient, only inpatient. The coarse to treat C-DIff is around 2-4 weeks. We have a few that the treatment is around 6-8 weeks.
Also we have several that are not responding to oral antibiotic treatment and the only other option is surgery. The surgery is a colectomy with a colostomy. If the case is severe enough the pateint could end up with a permenant colostomy for life.
So I say this simple procedure that can been performed as an outpatient with its high success rate and low cost is wonderful. I bet if you had this horrible infection you would jump at the chance to have this procedure.
rk900. How are you did you ever do the milkshake thing? Also how much liquid was with the enema and how long did you hold it in?
my mother has been in the hospital 5 times in the past 5 months, critically ill with c-diff. where in the viginia area can she go for treatment. we are in richmond.
Fascinating stuff! Thanks to all who have contributed in positive ways. I think I'm up next!
I'm a 12-year ulcerative colitis/crohns-colitis patient and I'm spending my Christmas in the hospital this year from an episode that sprang up much more suddenly and intensely than even I am accustomed to after starting a second round of stronger antibiotics prescribed to knock-out a sinus infection I've been struggling with. The C diff tests are coming back negative, but this is, as I understand it, OFTEN the case even when the bacteria is present.
Even if I don't have C diff caused colitis, there are several claims out there that this fecal transplant (or human probiotic infusion) procedure has had miraculous results in UC patients, so I'm pursuing it.
I don't know when I can expect to start this since I will be following up my hospital stay with oral prednisone and antibiotics until I have reached a normal-for-me-anyway state of health, intestinally speaking. I'm thinking it wouldn't be very useful to transplant bacteria into my colon while I'm taking antibiotics, but I don't think I'm going to let the gradually reducing dosage of prednisone postpone my start. In fact, I hope to shorten it. It's taking longer and longer with each UC episode, on average.
I just wanted to contribute to this post, seeing that enthusiasm for it seems to have ebbed, and state my intentions to pick up the torch and carry it now. Thanks again to all who have offered beneficial posts (you know who you are). For those interested, I'll continue to log my own experiences for your review.
If you attack me for attempting to share valuable information on something that could prove to be a relatively natural and potentially life changing process with others who have suffered as I have, please know that my mind is already made up that YOU are the idiot (you know who you are) and I will not engage.
I just spoke to the GI doctor on-call today at the hospital and mentioned the idea to him. He quickly discouraged the idea on the grounds that there were insufficient medical trials and resulting data and would be personally concerned with the possibility of contracting a disease from the donor, and that was the obvious concern, which simply falls to reason. Still, I'm not letting go of this that easily. It occurs to me that the medical machine largely conducts trials on medical opportunities to profit, and this wouldn't be one. It also occurs to me that there is an accumulating cost to my health with my current treatment, and my only other viable option is surgery. The risk seems reasonable, particularly since I've heard of no instances of bad outcomes. If you've heard of any, please post!
More to come . . .
Does anyone know of a Dr. or hospital in the Southern California area where they perform this treatment?
I just ran across this blog and comments. Have any of you guys been tested for gluten intolerance and/or celiac disease? My constant diarrhea and bloating normalized when I instituted a gluten free diet. I was serum negative for gliadin antibodies but my stool tested positive at Enterolab. I also had one of the main genes for celiac disease. My gastroenterologist wanted to do a biopsy to confirm a diagnosis of celiac disease, but I refused to do the gluten challenge. They don't ask a diabetic to consume sugary foods for weeks in order to confirm diabetes. They don't ask a recent heart attack victim to do a stress test in order to confirm a diagnosis of heart disease. Hard-line gastroenterologists are sceptical about the Enterolab tests, but I've found plenty of medical studies to back up the theory. And frankly, it just makes sense to look for antibodies in the stool, where they would be first formed, instead of waiting until you become serum positive. If you decide to go gluten free, you can't just reduce gluten, you must totally eliminate it from your diet - even a speck reinitiates the inflammatory process, even though you may not have symptoms. A good book to study on this subject is Healthier Without Wheat by Dr. Stephen Wagnan. Another is Celiac Disease: A Hidden Epidemic by Dr. Peter Green. Best of luck to you all in resolving your symptoms.
If you're thinking of doing a fecal transplant for crohn's, c. diff or whatever, it's better to use a sterile saline solution to mix up the poop than water. Water destroys some of the good bacteria. One doesn't need to get this done by others - you can do it in your own home. Just get some donor poo (I recommend a complete screening of the donor person for diseases)and mix it in a blender with saline solution until it is very watery and smooth. Strain it through a fine strainer into a container and then suck it up into a rectal syringe. Squeeze it in to your bum like a fleet enema. Leave it in for about an hour and a half. If you have trouble leaving it in, lie on your back and put two pillows under your bum, elevating it. This helps keep it in. Do this every second day for a few weeks and you'll be better!
My 94 year old grandmother is deathly ill with c. diff in NYC. She is in the hospital right now and on on a combination of flagyl and vancomycin for the past few weeks but with little effect. She still has severe diarrhea and might not survive. I mentioned fecal transplant to her infectious disease expert and gastroenterologist but they said "thats something that is done in Europe" and "we don't do that here" with a tone of contempt. I thought doctors are supposed to do every last thing that could possibly save a persons life. If fecal transplants have even a chance of success why deny it to an elderly lady as a last resort. Does anyone know if Medicare covers this procedure and if any doctors are currently doing this in New York City or nearby?
Any chance this procedure could be used to help cure excessive flatulence?
I did fecal transplantation about three years ago at home using my granddaughter's poo. It put me immediately into remission. I had to be on antibiotics again three times over the next two years. After the third time, I got another flare. That flare went away with probiotics and diet. I strongly recommend trying fecal transplantation if you have IBS, IBD, or especially if you have C-diff. I plan to do fecal transplantation again. I did not get any side effects from doing it, whereas the common side effects from traditional IBD medicines are very harmful.
I successfully did a self administered fecal transfer with my wife as donor. After 7 months of C diff and non working Vancomycin treatments I was desperate. It was immediately successful. Diahreah stopped at once. That was a year and a half ago. Gastro guy confirmed that C diff was gone. Doctors who refuse to do stool transplants should lose their license to practice medicine. It is cruel to pass up a cure when one exists!
Tara,
This technic is even better for those who has intestinal candidiasis. Intestinal candidiasis as a result of antibiotic intake is a chronic state because the friendly flora is gone. Candida overgrowth in the small bowel where the biggest concentration of Lactobacillus species live. Lactobacillus are very fragil and antibiotics destroy them . Candida destroy people health in a terrible way. I believe the only way to get endogenous bacterial strains in the gut is getting a fecal transplant. Probiotics colonize temporary the gut but they aren't able to replenish and implant the gut to get new colonies. The friendly flora is one of the most important things to be healthy. God bless this idea and I hope this become a regular treatment soon. I have 0 level of lactobacillus and bifidobacterias according to my last CDSA. In spite of heavy probiotic intake, my levels are 0. I have used all the best probiotics in the market including LGG, L. REUTERI, L. plantarum V-299, VSL-3, Probiotic 1, Primal Defense, PB-8, Dr, Osshira 12, etc. I have also taken tons of yogurt and kefir without result.
My main concern is the transport of the faecal matter. For someone to trasnport faeces in an ice cream container and not declare it is in contravention of International Air Trasnsport Association regulations on the trasnport of dangerous goods. Any body fluids are considered to be dangerous goods and must be declared and packaged according to strict guidelines and SHOULD never be transported in the passenger cabin of an airplane.
That woman who carried it on the plane would have been more than embarrassed if she had been subject to a search she would've been up for a hefty fine as well.
http://www.iata.org/Pages/default.aspx
Fascinating procedure - I've known about it but I am surprised not a word is mentioned about the remarkable yeast called Saccharomyces boulardii (sold as Florastor) which did wonders for me after I had horrible diarrhea due to antibiotics. It worked immediately (stopped having it the day after) and from what I understand, Florastor degrades toxins from C diffile thus reducing the virulence of it. You should try it before trying the fecal transplant and see if it helps. It also is believed to induce immune response to invasive microbes.
My 87 year old Mother has had C diff for over 4 months now, nothing has worked for her. She's been on the Vanco and Flagyl over and over again, this last time for 6 weeks, and still has diarrhea. She has been in and out of many hospitals and nursing homes but is now at her home with a 24/7 nurses aid, which is costing her $200 a day! She can't walk now and is totally bedridden and acts like she's getting diemntia. She was perfectly healthy before this hit her! Can someone please tell me where she can go to get this fecal transplant done near Detroit, Michigan? She is way too sick to travel far. She said she would try ANYTHING to get rid of this, it's the worse thing she has gone through in her life, and she's been through alot! Thank you!
I wonder if this treatment could also prime a person's immune system to resist local bacteria that cause illnesses like dysentery? If so, then maybe I could take a "poop pill" or chug down a cuppa chocolate feces before I eat the food and drink the water south of the border!
hi my husband has had stomach problems for 9 years every time its something else this is all throught out all the years 1. blastocystis (2004) hominus 2.GERD (2005) 3.helicobactor pylori (2005)4.high cholesterol(2005) 5.brain tumor fatty tumor(2006) 6.seizure(2007) 7. brain abscess( infection)(2007)8. ear infections through his life and headaches and migraines 9. gallblader removed(2007) 10.colitis(2008) er treated with 10 day antibotics was ok we could not afford the dr so never treated and he was fine 11. another colitis(2010) attack this time looking for answers we do not had insurance and need to find a way to have him better he has heartburn everyday takes prilosec everyday and has headache every day or every other day and takes excedrin migraines and it seams to keep him well enough but he is only 30 years old and has all ready been through hell . i don't know what else to do . just thought if any one else had any of the same problems would have so advice . i am trying to get coverage but with all pre excising conditions is so hard or not affordable .any way he was on strong iv antibiotics with the brain abscess 2x a day for 2 months he had pic line for it and the rx so now i see this could have caused some problems . it has been 2 years since colitis attack which he has bad stomach pain and vomiting and headache NO DIARRHEA so i am not to sure he has colitis as his symptoms are not that of colitis but he is a hard case with all the other issues this is er dx . any ideas would be helpful
thank you
Has anyone ever thought to use this treatment on children with autism? They often present with terrible GI issues. Perhaps their atypical neuro issues are related to horrible gut health?? I'm very curious to know if this has ever been tried with this population.
Okay i have IBD..and GAD (anxiety)... ulcertive colitis.. and ive talked to my doctor about this as well.. could this help me.."CURE".. and i understand one could die???? what are the chances??? anyone know or anything..
please help me out..
current flare up.. noush1960@yahoo.com
best regards
Laurie,
I have had C Diff and was treated in Sydney at the Centre for Digestive Diseases. I did an immense amount of research in to fecal transplants prior to going to Sydney and believe it is the only way to treat and cure C Diff. Prior to the fecal transplant or Human Probiotic Infusion as the CDD in Sydney calls it, I had courses of Flagyl and Vanco which did not cure the disease although the symptoms lessened whilst I was on them. I don't know anyone in Michigan but you could try the following: Dr Colleen Kelly in Rhode Island, Dr Lawrence Brandt in Montefiore Center, NY and the Duluth Clinic in Minnesota. There are not many people/ clinics doing this in the US, in fact not many in the world - I know as I contacted almost all of them! I hope this helps and you are able to get your mother better.
Dee,
It sounds to me like your husband has got some severe imbalances in the gut. My problems started over a year ago when I was diagnosed with Blastocystis Hominis. As a result of taking antibiotics to get rid of those plus a whole host of other antibiotics for other unrelated problems I contracted C Diff. Although I am not a doctor, having done a huge amount of research in to blasto, c diff I have learnt a lot about the gut and gut diseases. My feeling is that many of the problems you mention are related to the gut and your husband could do with a very comprehensive check up of everything to try and understand the root cause. The only place I know of is where I was treated... the Centre for Digestive Diseases in Sydney, run by Dr Tom Borody. He has pioneered a number of innovative treatments for diseases including several your husband suffers from.... Blasto, Helicobactor and Colitis. You will need to be persistent, it is very hard to get appointments as they are overwhelmed by calls from overseas. Borody knows his stuff and he should be able to help your husband.
I hope this helps
Faecal bacteriotherapy is hazardous in that infection from the donor could be transmitted to the patient and it involves delivery of faecal samples into the duodenum via a nasal probe. We propose to treat C. difficile using a modified FB, employing autologous samples collected from the patients prior to their treatment. Samples will be stored in a refrigerator and, should a patient develop C. diff., the samples are recovered, homoginised with saline and filtered. The filtrate is freeze dried and the resulting solid administered to the patient in enteric coated capsules.
does anyone know where fecal transplants are done
Paul Schleinitz, MD
Gastroenterology Consultants
2860 Creekside Cir
Medford, OR97504
Phone: (541) 779-8367
Fax: (541) 779-7471
Dr Schleinitz has done 11 fecal transplants. The hospitals where he has privilages have protocals and procedures in place for fecal transplants
By the way all 11 have returned to a lif without diarrhea. All are very pleased with the results. The stool transplant is usually given via colonoscopy. The stool cannot be more than 6 hours old, and not refrigated. Good Luck CDAD is a bad disease. Rob C
bloodtype and hla match not considered?
no immunosuppressive therapy as is the case with other transplants?
Laurie - My mother, 94, also has c-diff for the 3rd time since early August. I spoke with the infection specialist who is treating her in hospital today. He told me that there is an ifect. spec. at St.Joseph Mercy hosp. serving Ypsi & AA who does the fecal transplant. That is close to you
Mamac - it has been a long time since your last post. How is your daughter doing now?
Also, you mentioned that C. Diff testing often has a flase negative, is that true? When you say that it should be tested over 3 days, do you mean that there must be 3 separate stools over 3 days?
Also you say that you daughter had her gallbladder removed as well as a SoD operation, were these actually required or was she misdiagnosed?
my father has been suffering from c-diff for months and is totally reliant on diapers and house bound. It appears that it is getting very close to killing him. Our family is now looking into this proceedure with me, his daughter, as the donor....it would be a small miracle if it worked!
I contracted C-Diff almost a year ago. I have been hospitalized at least 5 times for life threatening symptoms relating to the infection. My symptoms have been minimized with the use of Vancomycin until recently. Now even with IVIG infusions and Vanco my symptoms are returning. Apparently fecal transplant or colon removal are my only options. Ethics committee's are stopping this procedure from being performed. Does anyone know where fecal transplants in Ontario or at least Canada are done. Minnesota is the closest state as far as the US is concerned. Any direct information would be appreciated
my father has been suffering from c-diff for months and is totally reliant on diapers and house bound. It appears that it is getting very close to killing him. Our family is now looking into this proceedure with me, his daughter, as the donor....it would be a small miracle if it worked!
Are there any places in the eastern US near NY city that perform this proceedure? Im suffering today without antibiotics from a relapse the second time with in a month.
Hello, I contracted C.Diff when I was in Hospital for a UTI. I am an almost 4 year Cancer survivor and was often on antibiotics for frequent UTI's. I had my Bladder removed in 2007, so I now have an illeaconduit. After diagnosis, I was on 2 courses of Flagyl then put on Vanc. It worked after a 21 day course, then I was symptom free for 8 weeks, then bam, hit again,long story short,I have been on Vanco since September and we are now testing donors for my transplant. Everytime,I tried tapering off Vanco,I would get down to 3 pills,and the symptoms would return. As far as testing the Donor, my Husband was tested and they discovered he carried H.Pylori with no symptoms! So I am now waiting on another to get results and go from there.I feel okay most of the time,but bowel movements are frequent with cramps and bloating, but much better on Vanco than not!I am so looking forward to my transplant and getting back to feeling normal again..Although, I don't know what that's like anymore. In answer to your question, I live in Toronto, and I know they are doing studies at Toronto General Hospital. I will definetley keep reading and posting, as this blog has helped me so much..It is so much better knowing that your ot going through this alone!
Sorry, I didn't clarify, that I received my C Diff in April last year. I really wish I had read this blog earlier, but I do want to say, Thank You to Tara, as my moto has always been "Knowledge is Power". While I don't believe everything I read on the net, it is fasinating how much I do find out and how much this information helps me. This is the most frustrating condition that I have ever been through, even though I had 10 hour surgery to remove my bladder..The not knowing what is happening and then trying to cure it to feel better. As I said previously, the Vanco has helped, but also,whatching what I eat has alot to do with feeling better as well. I was only 95lbs to begin with, when I first got sick I went down to 79lbs..So you can imagine I don't want to lose anymore weight. I am happy to say I am now 90lbs and very excited to get the transplant. Again, I cannot Thank you enough for everyone sharing their stories, it really gives us hope. Take Good Care Everyone, Kym
i just gave myself a shit transplant to see if it'll help.
i found a floater, mashed it up, and enema-ed myself. (first attempt with a spoon really didnt work)
i wonder if the girls from 2girls1cup could provide some tips on all this. they must have very healthy colons...
Maybe next time i just eat a 'dead soldier'.
when i was growing up i always had a hard time crapping. One year i visited Egypt (on the cheap / backpacker style) and was as sick as a dog for days. But for a few years after i was in tip-top health. Regular and easy movements and deminished asthma!
Then i received antibiotics for an ear infection, and i'm back to square one. So i'm looking into this 'transplanting'.
i dont think i'll try #147 - but it made me laugh.
The date has been set, I am having my Fecal Transplant, on March 4th, at Toronto East Hospital in Toronto..My Husband will be the Donor and I know it sounds crazy, but we are both so excited...I have been suffering since last April,been on Vanco since July... so I cannot tell you how much I am looking forward to this....I'll let you know! Kym
For Smithy...How is your BIL doing after his transplant,in Hamilton?
Negative comments must come, I can only assume, from people who do not suffer from these painful and debilitating bowel disorders. Anyone who is a sufferer must be at the very least intrigued by the idea that a cure could be available and most of them woiuld be willing to take part in any research project that was offered to them. Believe me I know.
I wonder if Stool Transplants would help Oxalate Issues? People that suffer from loosing there Oxalobacter formigenes. Oxalobacter formigenes are a very delcate GOOD Bacteria we have in our gut. It is the first to die off when taking antibiotics Flagyl is #1 Killer of Oxalobacter formigenes. When we lose the Oxalobacter formigenes the body can't process Oxalates from food. All food has Oxalates Vegtables spinch is one of the Highest Oxalates.
When our Body can't process Oxalates we get medical issues, the Oxalates go to inflamed areas in the body... Alot of Women tend to get Vaginitis and IC(bladder) issues because there body can't process Oxalates......
This may just help alot of Women that have Vaginitis :) and cure them so they can have a pain free life... THE VP FOUNDATION, and Oxalates Group at Yahoo great info.
I wonder if the Oxalobacter formigenes can be transfered with out dieing from the doner to the resiptiant??
Anyways thank you all for this blog...
Health and Happiness to all
My Mom has had c-diff three times so far. First time she was hospitalized with dehydration for four weeks and treated for c-diff. then two months later she contracted it again. Got tested two weeks ago and showed it gone. Last week it came back again after only one week. She has been referred to an infectious disease doctor and we are awaiting the appointment date. Maybe the stool transplant will help if they reccommend it. By the way she has Alzheimers and is almost 89 years old and before this hit her she was very healthy otherwise. pray for her recovery.
A concerned daughter.
ive read that young tortoises eat the poo (copraphagy) of other tortoises and it is thought that this is to build necessary fauna in their digestive systems. after i get over an illness i like to go out in the local park and eat a small piece of dog feces. i find this very helpful for my health. i dont eat a lot of it however. i think if you ate too much you might get sick.
Kymberley how did your fecal transplant go?
I'm on my third antibiotic for C-Dif after colorectal surgery. First Flagyl, then Vancomycin, now Xifaxan. I thought I was good after the Vanco but the C-Diff returned. I'm taking my first teps to a fecal implant. Question: Do you need to do a full prep (a la for a colonoscopy) before the procedure? Thanks.
I have been wondering of this procedure can be used to counteract the CACHEXIA found in cancer patients that may be brought on by the use of chemotherapy. If so, it would reduce the number of cancer patients - estimated at 20 to 40 percent - who die not of cancer but malnutrition.
Hi , Yes Id very much like to know how Kym's Transplant went, was it from the stomach or an enema.
Im thinking of having it done also, my flora was messed up from antibiotics and is inflammed. very sick most of the time.
Anyone in Australia interested in this procedure should contact Centre for digestive deseases in Sydney. Proffesor Borody and his team are at the for front of this procedure.
I have Crohns and a possible C-deff infection and will be going in for my first fecal transpalnt in about 5 weeks. I am actually looking forward to it. When you are in constant pain you become willing to try anything!
Hi, To George and Shane, I'm so sorry, for not posting...My transplant in March was via Enema and was not successful..:(.So I have been continuing on Vanco and a Probiotic since and have a transplant scheduled for August..This time via NG tube through to stomach!I have been managing pretty good, but cannot wait to have this over with!The problem with enemas is they don't reach all the way, and the little spores just can't wait to come out..After that procedure, I was really sick, as I had stopped the Vanco for a few days, hoping it would work!Thank God, the vancomycin works for me! Thank you for the concern and I promise I will update, probably early Sept. With all of the deaths going on in Niagara Region,I think they should seriously look into putting these procedures at the top of treatment list for C.Diff!
Bye For now, Keep Healthy :) Kym
Hey Everyone,
Well I had my transplant yesterday, it might be too soon to tell, but so far so good! I have been off the Vancomycin for over 24 hours and that in itself is a feat! For the past year, if I missed one dose, I would start the cramps, etc. I do want to share the fact that for the past 2 days before, I was terrified about the NG tube going in while awake, I mean terrifed, and believe me when I tell yo it was nothing, but a tad uncomfortable for a few minutes. I had it in for almost 3 hours and it wasn't a problem.So please do not let tales of horror stop you...Now just pray that it worked..I'll keep you posted..Take Care, :) Kym
Good Morning Everyone! :)
Kym Here, and it's day #4, with no vancomycin, no cramps and eating very normal! I cannot even believe it..I am so happy, I cannot tell you! I have suffered since April 2010, didn't even know what normal would feel like! Well, It's great, and I recommend this for all of the sufferers of this deadly Beast!!! You can also be reassured that I will be advocating up here in Ontario, for this treatment to be at the fore front for C.Diff patients!!!
I will continue to keep you posted....Healthy Thoughts to you all!
Kym :)
Hey, Me again, I just looked at some older posts and I wanted to reassure you all about something!
Donor Testing....
My Donor was screened for everything..Hiv,Hep C, H Pylori and anything else that could be found in the stool!So you wouldn't need to worry about catching diseases from said Donor, as they are extensively tested!
:) Kym
Kymberley, my father contacted C.Diff two and a half years ago after the last chemo session for lymphoma. Since then I was about to lose him nine times when he was admitted at emergency because of the C.Diff.
He is currently on flagyl antibiotic and it seemed like for some time it worked, until two weeks ago when he had the reoccurence two times already.
You mention that you had the Fecal Transplant, on March 4th, at Toronto East Hospital in Toronto. We live in Toronto and I am desperate to find a place that will do the fecal transplant.
Please pass me the contact information of the doctor that performs this treatement. I would very much appreciate it.
Thank you.
Kym, I was so excited to find this site and read your messages, that I actually started typing my previous message after only scanning through your and others messages.
I actually didn't even realize that you just had the procedure done and you already start feeling better. I read in the past that this is the only possible treatment for this bacteria, but as much as I tried I couldn't find anyone to perform it.
Please let me where should I call to get this procedure done for my father.
I'm very glad that you feel better.
Thank you and all the best.
Hi Stephanie,
I'm so sorry to hear about your Dad! If I can help I surely will, but I would rather private message, so my Dr. isn't overwhelmed.(Try and get him on Vancomycin in the meantime!)
It's Day 6 from the transplant and still feeling good, I do still find that I do have to watch what I eat right now...Just a little cramping when I eat certain foods!However,still no vancomycin and only once a day to the bathroom is such a luxury, and even nicer is the fact that my family is so relieved! That in itself is unbelievable!!
I will continue to post! Stephanie,write to me at
kymcal@hotmail.com and I will get back to you as soon as possible!
Keep Healthy Everyone! Kym :)
Hehehe..Me again..I just wanted to clarify something! I said I was eating normal again, and I am, except since about 6 months ago, even on meds, I couldn't eat cheese, eggs, or milk....Lactose intolerant right??? WELL...I could still go down to the Ice cream shop in town and eat a Banana split...Go Figure...Anyway, After the procedure, I tried a bit of cheese and it did give me mild cramps, so with what just happened in my body I am playing it safe and eating "Normal" for me!!!
Cheers :) Kym
STEPHANIE..Something is wrong with my email...For you Only, please call 416-305-1382....I really want to help, so I hope you read this blog!(Hopefully, no one else will use this??) I have no choice!
Kym
Dear Stephanie, I hope things are well, I am so frustrated as I wrote you a very long email, and then my account went down..courtesy of spam, I'm sure! I do want to help, so please call. Thanks Kym
Hello all, I just had to post to tell you all how wonderful I am feeling!!!!!It has been 9 days since my procedure and 9 days since vancomycin and all is very well! I cannot tell you how happy this makes me, not only for myself, but for my wonderful family who has gone through this hell with me! I will continue to take one day at a time...and eat those probiotics!!And... I will most certainley, in time, be crusading for this procedure to be available to EVERYBODY!!!!!!
Cheers Everyone, Stay Healthy, Kym :)
Hello all, Just posting to let you know that so far this transplant has been nothing short of a miracle for me! I am passed 2 weeks and haven't felt this healthy in over a year!
I so hope they can recognize this treatment at the forefront, this needs to be done EVERYWHERE!
Stay Healthy, Kym :)
Is there any place in the us that does fecal transplant my mom got c-diff from her job
Hi, I used fecal infusions to cure my son of Crohn's/Colitis and have now created a website where people can order guidebooks for doing fecal infusions at home. There is is different guidebook for several diseases and infections, including one for C. difficile. Infusions work very well and everyone, so far, who has followed the directions, has become well. The website is fecalinfusions.com. If you order a guidebook and you need help with anything, I will help you.
Hi all, I see the Dr. tomorrow, but it has been over a month since my transplant and I feel wonderful. To all of you out there, there is hope, just look for an Infection Control Doctor in your area!
Stay Healthy, Kym :)
Hi Sky, While I think your idea is fantastic, I couldn't get in (unless I buy the book!) to look over any of the details regarding a fecal transplant. Mine was quite simple, done through an Ng tube, this was my second try as the enema procedure, didn't go all the way! My Donor was tested for everything, hiv,h pylori, hep c etc..I'm sure I don't have to tell you the importance of that! I wish you luck,as you know, there is nothing like any of these diseases, and any hope of a normal life is wonderful!
Cheers, Kym
Hey Everybody,
To all of my Canadian Blog Friends.."Happy Thanksgiving"! Just wanted to check in and let you know..that I have past the 2 month mark. Yes, I had my transplant on Aug 5th and it is now Oct 10th and I am doing fantastic! I still cannot believe that such a simple procedure, literally gave me my life back! I am still planning to go forth with trying really hard to get this treatment to the forefront of the curing of C. Difficile. I will continue to update!
Good Health To Everyone!
Cheers, Kym
Does anyone know where I can get a stool transfer when I don't have C difficile, but I do have ulcerative colitis?
I heard that now this is being done in Hamilton, Ontario....
any information about this would be so appreciated!!
Thanks,
Pam
I would love to hear an update from Kimberley! How is it going?
Hi, my father, who lives in New Jersey, is on his fourth battle with C-Diff, within a 3 month period. The doctors have decided that he needs a fecal transplant, however, none of the doctors, here, have ever performed one. How can I find a doctor, who can either talk the doctors through the procedure or who can do the transplant, themselves?
Please help. My father is very malnourished and will not be able to handle a fifth go round!
Thank you,
Ilissa Gullickson
Illisa,
Your father doesn't need to see a doctor. He can just do the home remedy method and it is very effective. He can have the fecal transplant via enema using stool from a healthy donor . . . perhaps a spouse or even you can provide the healthy stool. Just purchase a three pack of enema containers (4.5 oz size is fine) from the drugstore. Mix a small amount of stool about half the length of your thumb in a blender with about 8 oz of .9 saline solution. He may need his doctor to call in a prescription for the .9 saline solution. It's often used for irrigating wounds and such, so the pharmacy will have it but a prescription is needed. Dump out the water that's already in the enema container as it contains a laxative. Mix the stool in a blender with the .9 saline solution and poor into the enema container. Insert the contents of the container in the rectum and hold at least a couple of hours, if possible. Then repeat the procedure every other day for 3 enemas. He will feel better very soon, likely within a week. Donor should be tested for c-diff and other parasites first. It is amazing how effective this procedure is! There is no better probiotic than the feces of a healthy donor. I suffered with c-diff for over 4 months, going through flagyl, vancomyacin, Xifaxin, and the just recently FDA approved Dificid. At the conclusion of the Dificid, and on the advise of my gastro, I performed this procedure using my husband's stool and it worked! My husband's stool had been previously tested for c-diff and other parasites and all was fine. When I started the Dificid, I asked my doctor, "how many rounds of antibiotics should I go through before I try the fecal transplant?". He said, "I would have done it the first time". That's how effective it is! When I was going through the procedure, I almost cried thinking what a low point this was in my life, mixing poo in a blender and inserting it in my rectum. But the real low point is the pain and suffering of c-diff for so many months. By the time we were on the third enema, we felt like experts and even managed to crack a few jokes. This was because I was already starting to feel a little better. My appetite had already started to come back. It's been 3 weeks and I'm still feeling good! I've started to gain back some weight. Next week I will go for testing and I'm looking forward to having 3 negative c-diff stool samples. Good luck to you and I hope your father feels better soon! I truly know what he has been going through.
Hi Debbie and all..It has been 3 and 1/2 months since my transplant..and...I am doing fantastic! I cannot tell all of you, I still marvel at the fact that this simple procedure, gave myself and my family back our life!My diet is nornmal, I have no more hint of that nasty bug in me! It was August 4th when I had my transplant, and honestly I was reborn! I continue to Pray for those in need, please, please be aggressive in trying to get this treatment, for you or your loved ones. I am almost ready to go forth and send my story to many Newspapers, I was waiting for time to pass, and to be really sure! I promise, everyone of you, that I will speak very LOUDLY on behalf of those who suffer and those who have passed with this horrible infliction! There is absolutley no reason, that Dr's are not using this treatment, as soon as it is discovered in a patient. I promise, I will keep you posted.
Keep Healthy, We'll talk soon,
Kym :)
Ilissa..Try to find a Doctor who specializes in Infectious Diseases, I know they are out there. I am so pleased, I was able to help someone on this blog, but I live in Canada, so I can't really help you, except to give you that advise.Try a big Clinic and ask specifically for an Infection Dr. I send you positive thoughts and strength. xo Kym
Kym,
Did you have some residual IBS symptoms for awhile? I feel like I fret over every little cramp or soft stool right now. I'm awaiting the results of my 3 stool samples dropped off this week, but I wish I had more confidence in a negative result. I worry that the fecal transplant by enemas, while initially so promising, might actually have not taken hold fully. Didn't you mention that you tried this first and it didn't work for you? Did you initially have some good results with the enema method and then after some period of time the c-diff returned? Just a couple of days after posting my original message, I began to second guess the results. I may just be over-reacting as my stomach and intestines have been through so much it make take some time to really feel "normal".
All 3 stool samples were negative for c-diff. Still not feeling 100% yet, though. Was hoping my appetite would be better and that I would have gained back more weight. Stools are solid though. It may just take some time to feel normal.
Debbie
Hi Debbie, Hey Hun, I hope this post finds you well, I'm sorry I just found this now!Yes, it will take you a little to feel normal again..your body has been through hell!!! I originally had a transplant through the enema and within 24 hours, I was sick as a dog, with C.Diff again, as I had been off the Vancomycin for 2 days before..So I went right back on the Vanco, until we could schedule another Transplant via NG tube. So from March until Aug 3rd, I took the meds, and then Aug 4th, I had the transplant..For about 2-3 weeks after, I would get cramps and some soft stool, and I would panic, but I hung in and I had no other testing, just felt better. It has been over 4 months now, and it's nothing short of a miracle. I feel great, stools are back to my normal, and I can now eat everything. For a while after, I watched dairy products, but now I am fine!
I feel very happy, for those of you who used the enema and got great results..I however, did not, and the Dr. explained, that going that way, the stool, doesn't always go all the way up!Also, I have to caution, that whatever Donor you chose, please make sure they are tested for everything! My husband was anxious to help, but they found H.Pylori in his blood, and god forbid we get that on top of the C.Diff..My Donor turned out to be a friend, who was tested for EVERYTHING..Hiv, Hep C, H. Pylori..etc...They don't fool around when putting someone else's bacteria into you. Just some useful info....Isn't this just amazing, though, no technology needed..just a blender...I send you strength my friend, and it sure sounds like your on your way...Please try not to worry about the weight, as with the C.Diff, I went down to 76lbs..I am now 89..so slowly but surely it's coming..(I only weighed 95 to start..hehe)Hears to your health Debbie...xo Kym
P.S. (I had bladder cancer and now have a iileaconduit..built by part of the instestine..so that could be the reason why my enema didn't work)..If you want you can email me at kymcal@hotmail.com and we can talk more freely!
Kym :)
If your husband has Cedif can he pass it to his wife?
Hey all, I answered last night but it didn't post..Anyway, I hope all had a Merry Christmas and are healthy!
Yes C.Diff is very contagious, only pure bleach can kill it...Almost 5 months and still going strong after my Fecal Transplant via NG tube...I hope everyone, keeps fighting for your health and find a Dr. to do this painless procedure...
I'm upset that my post last night was not put up...I take time to speak from my heart, in hopes to help people with information...
Take Good Care, and May 2012 bring you nothing but health and happiness...
Kym :)
Self-administered a transplant using my husband as a donor almost a month ago with amazing results! The key really was finding an enema bag with an occlusion bulb so I could hold in the transplant for 2 hours.
Everyone should check out the fecal bacteriotherapy group on Facebook and the new article "Swapping Germs" in Scientific American.
I have a lot of links and documents I'd love to share, but my comments aren't posted if they contain URLs. Any ideas for sharing documents?
Hi All, Just checking in, hopefully, this finds everyone well! I am still amazing, it's been over 5 months, and I am still amazing..Hoping to see this procedure, used by all the Doctors, to rid people of this horrible nasty disease.....
Interested to see how AJ is doing after your home transplant...I hope fantastic!!
Cheers to Everyone....
Kym :)
Another person dealing with C Diff. My heart goes out to you all more than I know how to say. I went into the Hospital for an amputation surgery and I left with this deadly C Diff. I'm on the Vancomin right now and hoping and praying this kicks it. Of course concern is there since so many people continue to battle this craziness. I know everything that happens has a purpose and when I get through this I can better help others do the same as well as inform people the dangers of this, since it's so unknown in my community and outside of it as well. No one has ever heard of C Diff. I hadn't either. I'm still awaiting my test results to know the extent of any of damage this has caused. I was told my colonoscopy and endoscopy looked great while they were in there. They found the c diff by taking a small sample from inside my small intestine. Not the norm for most cases from what I'm learning. Tends to be in the colon. So doing a fecal transplant into the colon, would not work for me is that I am assuming... Wouldn't I need to do this in a route that would entire my small intestine? And since I don't have a tube to go through my nose, would this mean creating capsules and needing to ingest my hubbies poop? So gross, but such a reality if this is what it comes too. They do not offer Fecal Transplant in very many places in the US and I am in Hawaii, where it's not even spoke of to be honest. Does anyone know of anywhere here or the closest state we could go to? Or do we just blend it up and capsule it at home? And how much and for how long would I need to do this? I have been through cancer, auto accident I was never to recover from or walk again from, countless surgeries, and I cannot tell you one thing, nothing has ever kicked my ass like C Diff. I was a very healthy 40yr old woman... Now I weigh 90lbs and thank god for my spirit and the need to show everyone I am ok... But I'm so not ok... This crap, literally needs to be gone and I dont eve want it to come back. The symptoms are insane and effect so much more than just my GI. I honestly told every doc week after week that I knew my body was feeling toxic, no one listened to me, not docs anyway. My family sure did. And thank god... Finally after 4 months I get the scopes and this is found. Hate having it, but grateful to get an answer and know im not loosing my mind. I imagine so many of you can relate... Please anyone with some answers that can help this non crazy woman in Hawaii who's considering ingesting her hubbies poop....Aloha and wishing you all good health, peace and so much unconditional love in this world.
Oh Jay, Wow, Your Story is incredible...I send you Hugs and Strength...I'm sure you've read my story, I too have fought Bladder Cancer(bladder removed) and skin Cancer,but nothing like this C.Diff! My heart goes out to you!
I was on Vancomycin for 14 months..yes, that's what I said, everytime I would taper off, the symptoms would come back..I only weighed 95lbs and I went down to 70..(but I too am a very positive person with alot of fight in me..)Anyway, I found an Infectious Disease Dr. and he did the transplant...I have been completley symptom free and off Vanco since Aug 4th...I had the transplant through the NG tube, but maybe the capsules would work!!! An enema didn't work with me, it didn't go all the way up, maybe because of scar tissue..I have no bladder, so my intestines were cut to make an ileaconduit..In any case, it's worth the process, as I've learned, once you have the bad bacteria, they have spores that hide, and can crop up at anytime!
So please my Dear Jay, don't give up hope..keep looking and def look into the capsules, although I'm sure there is some Dr that would do it!
I will send positive thoughts for you my Dear, and know there is hope, you will find it!! Keep me posted..
Kym :)Ridgeway, Ontario
Hi,
Is there anyone who can provide some info of where this can be done in Europe ?
Although I am ready to come to the US if anyone can help me get this transplant done.
David
The study states: "Select a stool specimen (preferably a soft specimen) with a weight of 30 g or a volume of 2 cm^3."
However, if one assumes human stool has a mass density of approximately 1 g/(cm^3), then this sentence does not make sense. A mass of 30 g is much more than one would expect of a stool sample of 2 cm^3, which would be 2 g. So from the description, it is not clear to me how much stool needs to be used. Can anyone explain?
(I am thinking of using empty 000 gelatin capsules; I can't imaging why someone would want to use a milkshake (yuck!).)
Hi. Do you know of any physician in the Kansas City area that does stool transfers?
What generally do they cost?
Thanks
John Hansen majorhart@sbcglobal.net
Hi. Do you know of any physician in the Kansas City area that does stool transfers?
What generally do they cost?
Thanks
John Hansen majorhart@sbcglobal.net
I think that we will see more information about C-Def and it's bad effects when more Gut Doctors get sued for patients killing them selves because of the depression it causes. There are no studies on this yet but some good Malpractice lawsuits for C-Def suicides would get Gastroenterologists off their ass to get the word out. I lost a loved one... the C-Def drove them over the edge and I am collecting material for all of you to sue!
Rod
Rod, I am so sorry about your loss. Yes, It is an incredibly horrific disease..The sad part is that if every Doctor did this procedure, these kinds of things wouldn't happen. I had my transplant via Ng Tube last August 5th, so I have been fine for over 8 months. To think of such a simple procedure, lasting all of 20 mins could cure....I too suffered for over a year and a half, along with my family. I wish you luck in your endeavours and send you strength everyday.
Kymberley
I've been suffering with this for 30 years. Where can I get the transplant done in the U.S.?
Has anyone had a fecal transplant at any hospitals in NC?
Also, is there a higher success rate with the nasal tube as opposed to the colonoscopy route? Thank you so much, everyone for all the posts on your c-dif experiences. Trying to learn all I can. Weary of this disease!
If you have read this far, you may find bacteriophages of interest:
http://en.wikipedia.org/wiki/Bacteriophage
Some excerpts from the wikipedia article:
A bacteriophage (from 'bacteria' and Greek φαγεῖν phagein "to devour") is any one of a number of viruses that infect bacteria.
They have been used for over 90 years as an alternative to antibiotics in the former Soviet Union and Eastern Europe, as well as in France. They are seen as a possible therapy against multi-drug-resistant strains of many bacteria.
Phages were discovered to be antibacterial agents and were used in the United States and Europe during the 1920s and 1930s for treating bacterial infections. They had widespread use, including treating soldiers in the Red Army. However, they were abandoned for general use in the West for several reasons:
- Medical trials were carried out, but a basic lack of understanding of phages made these invalid.
- Phage therapy was seen as untrustworthy, because many of the trials were conducted on totally unrelated diseases such as allergies and viral infections.
- Antibiotics were discovered and marketed widely. They were easier to make, store and to prescribe.
- Russian research continued, but was published in Russian or Georgian, and was unavailable internationally for many years.
Their use has continued since the end of the Cold War in Georgia and elsewhere in Eastern Europe. A monograph written by Nina Chanishvili in 2009, in Tbilisi, Georgia gave a thorough analysis of the results of phage therapy.
Hi Everybody, Just wanted to come on and let you know, that today it's been an entire year since my transplant via NG tube. What a success story, it was such a long haul...To GGB, I originally tried the enema route, to no avail. I had the NG tube, that took approx 20 mins, with a couple of hours relaxing after, and I have never looked back! I was on Vancomycin for over one year, and the night before my procedure, was my last capsule..(Aug,4th,2011). I pray for anyone that is suffering from this to please..please..keep looking for a Dr. who will do this...To this day, I cannot understand WHY, this treatment is not at the forefront of this Horrible disease! I lived in Toronto,Ontario, when I came down with this, and was treated there. I thank God everyday for this cure! I will continue to send strength to those who are suffering. Please keep fighting, if you can!! Love and Hugs..Kym
..Also, I just read some past's post, and felt I should add..I do not have an NG tube...One was inserted just to do this procedure..Just thought you should know this! It is very simple and painless, just sounds scary. All I know, is that it worked, and almost instantly!!!
Keep Healthy :) Kym
just an fyi -
dr. brandt of the montefiore hospital in the bronx, ny is does
fecal transplants and is currently enrolling people for an
nih clinical trial/ study on the treatment.
http://www.youtube.com/watch?v=JRZxOU1x71c
http://www.einstein.yu.edu/departments/medicine/divisions/gastroenterol…
A fecal transplant healed me from coliltis in October 2012 after 3 1/2 years of suffering.
Read my blog -http://healed-from-uc.blogspot.com
It WORKS!
I am symptom free and drug free!!!
Hi Everybody..It's been a longtime, since I've written on here...but just wanted you to know..That my Fecal Transplant was August 5 2011..and I have been Healthy ever since..No Vanco, after having to be on it for a year and a half...I was very lucky...I hope everyone is healthy and has had success like I have...Life is good! Take Care and Keep Smiling Kym :)