tags: researchblogging.org, speciation, adaptive radiation, , diversification, ecological opportunity, community assembly, species interactions, North American wood-warblers, Dendroica species, Daniel L. Rabosky
Yellow-rumped warbler, Dendroica coronata, After Hatch Year male.
Several questions that motivate my own research and thinking are; How do species arise? Where does all this biodiversity that we see on Earth come from? Does speciation occur as a series of slow and gradual accumulated changes or is it an explosive process that occurs within a short period of time, followed by long periods of evolutionary quiescence? These are a few of the most fundamental and contentious questions in evolutionary biology today.
The fossil record suggests that much of the biodiversity we see arose quickly in response to ecological opportunities: abundant resources combined with few, or no, potential competitors. As the niche became more crowded, the rate of speciation decreased. This process is known as density-dependent diversification. But can we learn about the tempo of adaptive radiation for the vast majority of species groups for which there is no fossil record?
Cornell University evolutionary biology graduate student, Daniel Rabosky, and his advisor, ornithologist Irby Lovette, wondered the same thing. Evidence from previous DNA analyses suggested that speciation events followed the fossil record model of rapid diversification followed by a general slowing of diversification, but this hypothesis had never been tested at the species level.
Rabosky and Lovette developed a mathematical test that distinguishes density dependence patterns of speciation -- the way that closely related species divide up their environment -- from alternative explanations. According to this ecological opportunity model, when there are few relatives around to compete for resources, such as when a habitat is first colonized, species differentiate rapidly. But as more species evolve to fill available niches, competition becomes fiercer and provides fewer opportunities for new species to arise. Rabosky and Lovette tested this model using a family tree that they constructed from the DNA from New World wood warblers within the species-rich genus, Dendroica.
"Right after the dinosaurs went extinct, most people are aware that there was a huge explosion of mammal diversity," Rabosky points out. "That extinction created lots of ecological 'space' -- opportunities for new species of mammals to take advantage of resources. On a much smaller scale of both space and time, this little group of warblers from North America might be playing out the same story."
For example, an initial burst of rapid speciation is a pattern that has been observed repeatedly for island species, such as "Darwin's finches" of the Galapagos Islands, but species that experience a period of explosive adaptive radiation on large continental landmasses are thought to be very unusual.
"What's interesting is that this happened on a continent where it's ecologically much more complex than an island," Rabosky reports. "You've got lots of competitors. Clearly, lots of other birds were already here, eating insects and probably doing what warblers do now."
Previous work reveals that Dendroica warblers are a classic example of behavioral niche differentiation, where species that evolved together show subtle differences in their foraging and breeding behaviors [DOI: 10.1371/journal.pbio.0060071]. Additionally, reconstruction of a time-calibrated mitochondrial DNA (mtDNA) phylogeny revealed that Dendroica underwent an explosive burst of radiation early in their evolutionary history, followed by a pronounced decline in their rate of overall diversification. Thus, the observed tempo of speciation among Dendroica appear to be related to ecological opportunism tempered with complex interactions between nascent species.
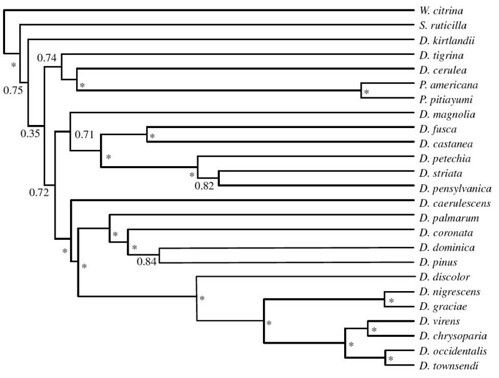
To test whether the ecological opportunism model was consistent with observations of speciation in wood warblers, Rabosky and Lovette first reconstructed a molecular phylogeny for all Dendroica and four additional wood warbler species that fall within this clade using data from six mitochondrial genes and six nuclear introns. This provided a detailed picture of the genetic relationships between all the species within this clade and the sequential order when they evolved (figure 1);
Figure 1. Maximum clade credibility (MCC) tree from Bayesian analysis of all continental North American Dendroica wood warbler species. Nodes marked with asterisks are supported by posterior probabilities of more than 0.95. Tree is based on more than 9 kb of mtDNA and nuclear intron sequence. Branch lengths are proportional to absolute time [larger view].
DOI: 10.1098/rspb.2008.0630.
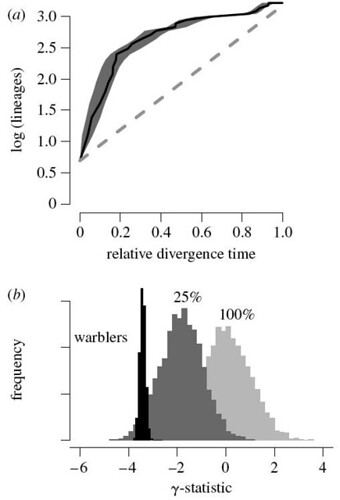
This phylogeny supported previous findings that, after an initial explosive burst of speciation, Dendroica divergence rates slowed dramatically through time. When analyzed mathematically, this initial rapid radiation is clearly revealed by a lineage-through-time (LTT) graph (figure 2a);
Figure 2. (a) Log-lineage through time (LTT) plot for North American wood warblers. Black line indicates LTT curve for the MCC tree (figure 1), and grey shading indicates 95% quantiles on the number of lineages at any point in time as inferred from the posterior distribution of phylogenetic trees sampled with MCMC. The dashed line indicates expected rate of lineage accumulation under constant-rate diversification with no extinction. Lineages accumulate quickly in the early phases of the radiation relative to the constant-rate diversification model. (b) Posterior distribution of the γ-statistic for wood warblers (black) in comparison with the corresponding null distributions, assuming either complete (f=1) or incomplete (f=0.25) sampling. Negative values of γ relative to the null distribution indicate decelerating diversification through time [larger view].
DOI: 10.1098/rspb.2008.0630.
Figure 2b describes the numbers obtained for the γ statistic tests for slowdowns in the speciation rate. This statistic analyzes the distribution of internode distances through time. A γ value less than -1.645 indicates there are highly significant (pDendroica species, they found that their γ test was still valid, indicating that this genus experienced an explosive bout of adaptive radiation before settling down to a "more normal" rate of speciation that is approaching the background tempo.
This test is an incisive tool that provides a clearer understanding of the pattern and rate of speciation for groups of closely related species, even in the absence of a fossil record, by analyzing their DNA. Rabosky and Lovette's findings suggest that the rate of Dendroica speciation was mediated by ecological interactions among species and that lineage and ecological diversification in this group are interrelated, as predicted under the ecological opportunity model. Their research also suggests that an initially rapid rate of adaptive radiation might not be specific to islands; it might be a more universal process than originally thought among for species that arose on major landmasses, particularly in the wake of a major extinction event.
Sources
Rabosky, D.L., Lovette, I.J. (2008). Density-dependent diversification in North American wood warblers. Proceedings of the Royal Society B: Biological Sciences, -1(-1), -1--1. DOI: 10.1098/rspb.2008.0630.
Cornell University press release (quotes).
Also read
Phillimore, A.B., Price, T.D. (2008). Density-Dependent Cladogenesis in Birds. PLoS Biology, 6(3), e71. DOI: 10.1371/journal.pbio.0060071.
- Log in to post comments


What is the "relative divergence time" in figure 2a? Are we talking about a relatively recent pleistocene radiation, or something much earlier?
This statement caught my eye, "According to this model, when there are few relatives around to compete for resources, such as when a habitat is first colonized, species differentiate rapidly."
This sounds like a sympatric speciation model to me? It implies that a habitat is colonized and then the original colonizer somehow speciates into multiple species in the same habitat or geographical area. Is that what these authors are advocating? If so, call me skeptical.
Joseph - does it need to be a sympatric speciation model? What about just a greater tendency to proceed to speciation during allopatry, due to the many possible niches each could fill as they colonize. When there are many relatives around, then you would be less likely to speciate in allopatry because you're more constrained.
Just my hypothesizing, I don't actually know what the authors propose on this subject.
two questions:
1) the pedantic question: since "P. pitiyaiumi" and "P. americana" are nested within Dendroica, does this make Dendroica polphyletic and invalidate it as a clade? (Just love to slap Hennigians with this challenge!)
2) the serious question: this explosive burst of diversification reminds me (us? because of the references in the paper to island biogeography) of, well island biogeography. What is the origin of Dendroica? Might this initial burst represent the consequence of a colonization event? (Alien species fortuitously 'introduced' to a novel environment... i.e. Where are Dendroica's nearest relatives?)