tags: evolution, evolutionary biology, behavioral ecology, animal behavior, molecular ecology, parental care, mating systems, monogamy, sexual selection, frogs, poison dart frogs, Dendrobatidae, Ranitomeya, researchblogging.org,peer-reviewed research, peer-reviewed paper, journal club
To know the breeding system is to know the genetic architecture of a species.
To know the evolution of a breeding system is to know how evolution works ..
~ Lewis & Crowe, Evolution (1955)
Genetic tests have revealed the secret sex life of a tiny poison dart frog species that lives in the Peruvian rain forests: remarkably, it turns out that these frogs are monogamous. But the reason this species is monogamous is surprising: it's all about the size of the pools that their tadpoles mature in. This is the best evidence yet that just a single cause can affect evolution of a major life history trait, such as a species' mating system.
The forces that underlie the evolution of biparental care have been the subject of lively debate within the scientific community, and in turn, how different forms of parental care affect the evolution of each species' mating system is also not well understood.
According to one long-standing hypothesis proposed by mammologist Devra Kleiman, monogamy may arise as a life history strategy when biparental care becomes critical to offspring survival. In this situation, both parents may experience higher reproductive success by investing in their mutual offspring instead of seeking extrapair reproductive opportunities [DOI: 10.1086/409721]. Life history studies of a variety of avian and mammalian taxa support this hypothesis.
But which ecological factors drive the evolution of biparental care and monogamy? Is just one ecological factor enough, or is more than one necessary? One way of answering these questions is to compare the life histories and ecologies of closely related species where one species is monogamous and provides biparental care while the other is promiscuous and does not provide biparental care. But which species fulfill these criteria?
Jason L. Brown, now a postdoctoral researcher at Duke University, spent six years studying Peruvian poison frogs while a graduate student at East Carolina University. In those studies, he evaluated key ecological and behavioral differences between Peruvian poison frogs and framed those key differences in the context of differing parental care strategies. Building upon his dissertation work, Dr Brown and his colleagues, Victor Morales, at the Ricardo Palma University's Natural History Museum in Lima, Peru, and his dissertation advisor, Kyle Summers, Associate Professor in the Department of Biology at East Carolina University, took a closer look at two closely-related poison frog species: the mimic poison frog, R. imitator, which exhibits biparental care (where both parents care for their tadpoles for a period of months) and the variable poison frog, R. variabilis, which exhibits male parental care (where only the males transport tadpoles to a pool of water and leaves them to fend for themselves).
Like all amphibians, poison frogs are intimately bound to water because that is where they lay their eggs and where their tadpoles mature. But bodies of water are filled with numerous hungry mouths, so the pressures of predation pushed the tiny poison frogs out of the larger ponds and into the considerably smaller (but safer) pools contained in the cup-shaped leaf axils of a variety of plant species that grow in trees. These tree-top plants are not often visited by hungry predators, but their small pools of rainwater, known as phytotelms, lack sufficient nutrients to successfully grow a tadpole (unlike larger ponds of water).
To ensure that their tiny offspring grew up into mature frogs, the mimic poison frogs adopted a new tactic. Merely transporting their tadpoles to individual phytotelms and abandoning them to grow up on their own was no longer enough: the males needed help. Female mimic poison frogs evolved a behavioral strategy known as trophic egg feeding, where they lay unfertilized nutrient-rich eggs into each tadpole's phytotelm for it to eat.
But this strategy requires teamwork: while the female spends her days eating voraciously so her body can manufacture these special trophic eggs, the male visits each tadpole every day or two, for only he knows where they are hidden. Whenever a tadpole nibbles on its father, the male calls to his mate. The female responds to the male's calls by following him to their offspring's phytotelm, jumps into the tiny pool and produces a trophic egg, which the tadpole consumes. The female produces at least one trophic egg for each tadpole every week for a period of months (Figure 1):
Figure 1: Biparental care in Ranitomeya imitator. The male leads a female to a deposition site within their territory. The female will then oviposit one to four eggs on the surface of a leaf, away from water. A, A week after oviposition, the male returns and retrieves its tadpoles, breaking the tadpole free from the egg sack and allowing the tadpole to wriggle onto its back. B, The male then deposits the tadpole in an unoccupied pool of water within the pair's territory. The male will then survey the pool every couple of days and determine when the tadpole is fed trophic egg(s). C, To initiate egg feeding, the male begins to call from the surface of the pool, and after a short period, the tadpole's mother arrives. D, After an extended bout of calling, eventually the female will dive into the pool and deposit a trophic food egg (depicted in white within the center of the plant; female is within the pool). This behavior is repeated on average every 7.3 days throughout the duration of development of the tadpole. [larger view]
DOI: 10.1086/650727.
Dr Brown's dissertation work revealed that the closely related variable poison frogs did not engage in biparental care, despite sharing the same habitat and habits as the mimic poison frog (The mimic poison frog imitates the brilliant color patterns of this poisonous species, so they even look very similar). In variable poison frogs, only the males relocated newly hatched tadpoles to a phytotelm and left them there to mature. But what was different between these two species?
To investigate this question, Dr Brown and his colleagues compiled a variety of natural history data from 404 frog species distributed across the entire amphibian taxonomic order Anura and reconstructed the evolution of these characters. They then analyzed these data to find any correlations between the evolutionary transitions to phytotelm breeding and parental care (Figure 2):

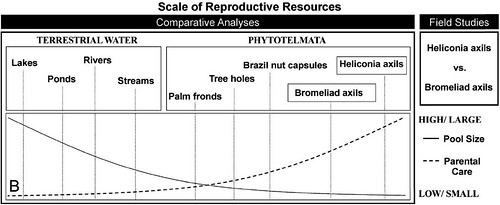
Figure 2: Different scales of this study. Comparative analyses were carried out on a much larger scale than our experimental and genetic analyses both in terms of phylogenetic scope (A; looking at the order Anura vs. two closely related species in the genus Ranitomeya, family Dendrobatidae) and in the context of type of reproductive resources (B; comparing all sizes of reproductive resources being used vs. two small phytotelmata). In general, species that utilize larger bodies of water exhibit lower levels of parental care.
DOI: 10.1086/650727.
The only difference that Dr Brown found between the variable and mimic poison frogs was the size of the phytotelms themselves: male variable poison frogs deposited their tadpoles into pools that averaged 112 mL in volume, compared with just 24 mL in those pools used by mimic poison frogs -- a five-fold size difference. Based on these data, Dr Brown and his colleagues proposed that pool size was the driving force in the evolution of biparental care. The team then tested their hypothesis by moving tadpoles from both species into differently sized pools and monitoring their growth (Figure 5):
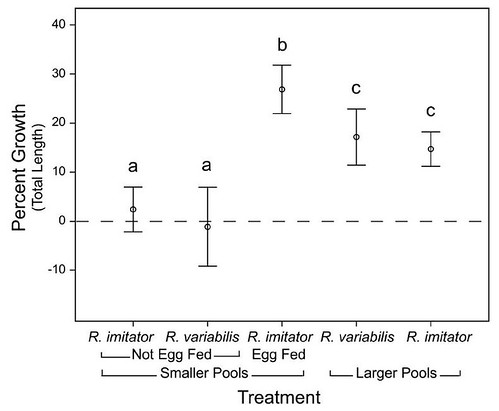
Figure 5: Reciprocal transplant experiments. We used five treatments in which the tadpoles of each species were placed in natural phytotelmata and allowed to consume the resources within the pools. The tadpoles were weighed and measured at the start and end of the experiment. Error bars = 2 SEs.
DOI: 10.1086/650727.
The team found that tadpoles in larger pools thrived while tadpoles in smaller pools did not grow. This led them to conclude that tadpoles maturing in larger, more nutrient-rich pools didn't need both parents, unlike their smaller-pond counterparts. Thus, species that raised their tadpoles in smaller ponds required attention from both parents. In turn, this favored parents who remained devoted only to the offspring that they produced together -- but did it also drive the evolution of monogamy?
To answer this question, the researchers first had to determine whether mimic poison frogs are truly monogamous. They sampled DNA from 12 mimic poison frog families and their tadpoles and were surprised to find that in 11 of those frog families, the parents were faithful to one another, while in only one family did the male frog have two mates. This is even more surprising because genetic testing has shown that many animals thought to be monogamous are less faithful than previously believed.
Monogamy "turns out to be relatively rare, even in birds and mammals -- particularly in mammals -- and reptiles," reported Dr Summers. "Finding a frog that has a monogamous mating system was pretty novel for us."
In contrast, genetic studies of 12 pairs of the closely related variable poison frog indicates that this species is promiscuous.
"Others have found evidence of social monogamy in amphibians where parents remain paired, however they didn't look at the genetics of these couples and their offspring to confirm this," explained Dr Brown. "Or they have looked at the genetics and observed that they are actually promiscuous."
But which came first; parental care or small pond size? The team explored this question by estimating the rates of evolutionary change for all four possible combinations of these two character traits, each with two possible states: terrestrial water body breeding versus phytotelm breeding and no parental care versus parental care. Their analysis indicated that the most likely evolutionary pathway from ancestral pond-breeding species without parental care to phytotelm-breeding species with parental care progressed from pond breeding to phytotelm breeding, followed by the evolution of parental care (Figure 3):
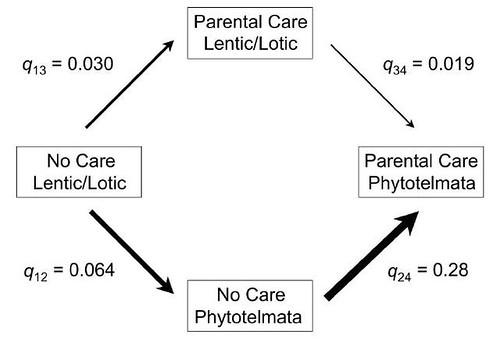
Figure 3: Evolutionary pathways from the ancestral condition of terrestrial water body (lentic/lotic) breeding without parental care to phytotelm breeding with parental care. Each value of q represents the rate estimated (via maximum likelihood) for that particular transition from our phylogenetic tree (Supertree [Nexus], available in the zip file in the online edition of the American Naturalist). Simultaneous changes in both characters are not allowed under the maximum likelihood model employed in the method. Log-likelihood ratio tests reveal that the most likely evolutionary pathway involves the evolution of phytotelm breeding followed by the evolution of parental care.
DOI: 10.1086/650727.
The team also investigated the interaction between trophic egg feeding and biparental care to determine which of these characters evolved first. To do this, they mapped those characters onto a "mirror tree." A mirror tree is a graphically elegant analytic method where particular character states are mapped onto a phylogenetic tree to test the evolutionary interaction between those particular characters. The underlying assumption of mirror trees is that these interacting characters co-evolved (Figure 4):
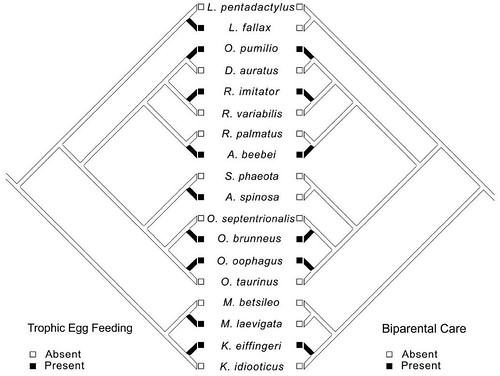
Figure 4: Mirror tree including only those species with egg feeding and one closely related taxon, illustrating the concentrated changes test of an association between egg feeding and biparental care (see Dryad data; Breeding Site Refs, Supertree [Nexus], PhytoEvol, and PCEvol are available in a zip file in the online edition of the American Naturalist).
DOI: 10.1086/650727.
This analytic method reveals that trophic egg feeding evolved before biparental care.
Overall, the researchers think they have found convincing evidence of an evolutionary chain of causation: first, by dramatically reducing the size of their breeding pools, mimic poison frog couples were forced to work together, providing biparental care for their offspring so the tadpoles would survive and mature. That change then culminated in both social and genetic monogamy.
"This is the first discovery of a truly monogamous amphibian," said Dr Brown. "These frogs are truly devoted to their offspring, and to each other."
Is it possible to draw any parallels between mimic poison frogs and human behaviors?
"People are interested in whether there are parallels between mating systems of other species and our own. Of course, the human situation is so different from other species. It's somewhat perilous to over interpret the similarities," Dr Brown wisely cautioned. "You can't just translate it."
Dr Brown's fascinating research into the reproductive habits of mimic poison frogs was filmed by the BBC and included in their natural history documentary series Life in Cold Blood, narrated by David Attenborough:
Source:
Brown JL, Morales V, & Summers K (2010). A key ecological trait drove the evolution of biparental care and monogamy in an amphibian. The American Naturalist, 175 (4), 436-46 DOI: 10.1086/650727; PMID: 20180700
Read more about Jason Brown's fascinating work in Poison Dart frogs (index of PDF links to most of his published work).
- Log in to post comments




Thanks -- this is great and I will use it in my animal behavior class!
oh, wow! i am surprised and pleased that you dropped in to read this essay! i actually have pitched an idea to write an essay (or maybe a series of essays) in a story-like format (similar to this essay) about the evolution of the red queen hypothesis. i imagine that you, in particular, will have some interesting things to contribute to that .. !