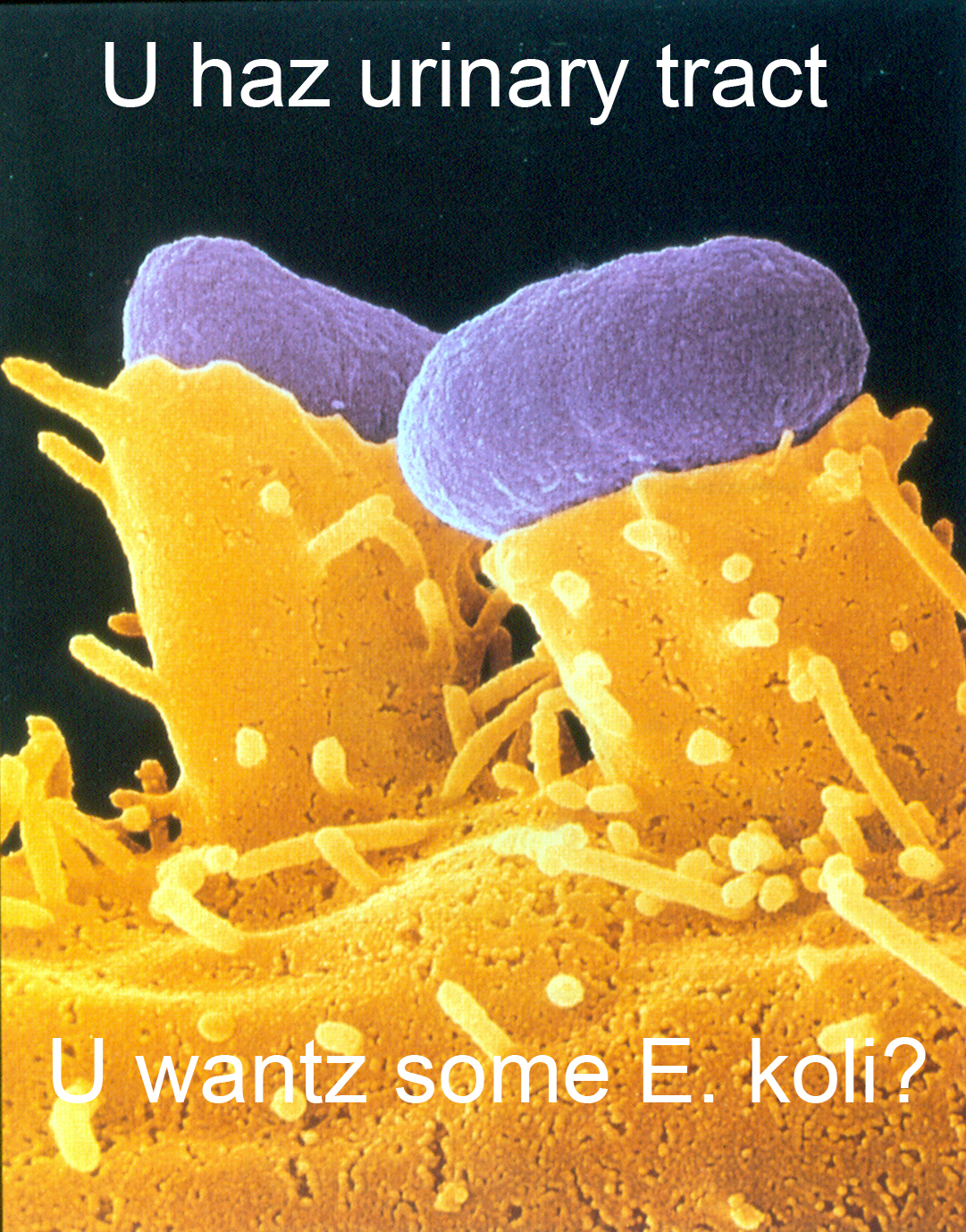
Yes, this is O157:H7, not ExPEC. Bully for you.
One thing regarding popular accounts of antibiotic resistance I've noticed is that there is an overemphasis on the evolution of resistance, and an underemphasis on the spread of resistant bacteria. While the evolution of resistance is important, most of what we see in a hospital is not the de novo change of a sensitive strain into a resistant one (i.e., evolution), but, instead, the survival and spread of already resistant bacteria in this antibiotic-laden environment.
What this means is that changes in the frequency of resistant organisms, for better or for worse, depend not only on antibiotic use, but also on other ecological dynamics--that is, because of clonal spread. In other words, a resistant strain might not become more common because it is resistant, but because it does something else better than most other strains (e.g., it might be hard for the immune system to recognize and attack it).
A recent paper about antibiotic resistance in E. coli that cause urinary tract infections ('UTI') indicates that clonal spread is driving antibiotic resistance. The researchers collected a total of 780 isolates of E. coli from every referred patient at a university health center with UTI during a 14-week period for four consecutive years.
What they found was that there was no trend (up or down) in the frequency of resistance to trimethoprim-sulfamethoxazole, ciprofloxacin, cephalothin, or nitrofurantoin. Only ampicillin (amoxicillin) decreased significantly from year three to year four. Shortly before the study began, the health center switched treatment of UTI from trimethoprim-sulfamethoxazole to either nitrofurantoin or ciprofloxacin, yet the prevalence of resistance to all three of these drugs remained unchanged.
The authors also looked at the genetics of these strains. Most of the UTI were caused by a limited number of genotypes (genetic lineages), some of which are drug resistant. The odds of women without common exposures would be infected with this limited subset of genotypes is quite small. In fact, if drug use were generating novel resistant strains, we would expect to see many more resistant genotypes. Instead, what we see is the 'sorting' of pre-existing resistant UTI E. coli, and this sorting appears to be independent of antibiotic use.
So what does this mean for public health policy?
Trying to treat a particular problem, such as a drug resistant UTI-causing E. coli will not work in isolation. Like within-household transfer, where it might be necessary to treat asymptomatic family members and pets (who carry these resistant organisms), we have to 'embiggen' (to use Madame Wev's phrase) the scale on which we envision the problem--this is not a problem that starts at the hospital door. We also need to determine what the reservoirs of these resistant UTI strains are (i.e., where else they live), and attempt to limit the spread from those reservoirs.
- Log in to post comments

Oops, I think that you need to rethink your wording of that opening paragraph. If evolution is the change in allele frequencies in a population, then clonal spread is evolution. The distinction you mean to make is that population accounts overemphasize the origin of resistant strains, and underemphasize their spread.
Dan,
Nope. The reason I refer to clonal spread as an ecological problem is that "the change in allele frequencies in a population" implicitly assumes that over generational timescales, alleles do not cosegregate at a genome-wide scale. In a population composed of organisms, like bacteria, that are essentially 'super-alleles' (again, on a generational timescale), the spread of resistant genotypes should be seen as a migration issue--that is an ecological problem--and not a phenomenon of the generation of novel genotypes (either by mutation or allelic recombination) which I would classify as an evolutionary problem in this context.
This is why I repeatedly write about how any evolutionary theory that does not take into account 90% of the earth's biomass, and most of its genetic diversity needs a little improvement.
Mike, I'm baffled by why you consider migration -- one of the four fundamental population genetic forces -- to not be an issue of evolution. This is not to say that it is not also and ecological issue. I'm sure you are the last to want to decouple evolution from ecology.
But leaving that aside, you believe that evolutionary theory has shortcomings. Before any progress can be made on those shortcomings, isn't the first step to identify this as a problem that is/should be within the bounds of evolutionary theory?
Dan,
Historically, microbial population biologists have modeled the spread of bacterial genotypes using ecological theory. This is because hosts typically are treated as demes, and thus will contain only one or a few genotypes within a species. In other words, when E. coli strain A replaces E. coli strain B within a person, I (and most of my colleagues) treat that in the same way ecologists would treat competition between two species. Is this also genetic change within a population? Sure, but I don't think viewing it from this perspective is as useful as thinking about it as an ecological problem.
The deeper issue has to do with diversification that can lead to speciation. In eukaryotes (which are the subjects of most population genetics theory), migration is an issue because migration, through genetic introgression, can break apart genetic diversification, and, thus, retard (or inhibit) speciation. In bacteria, migration and the resulting genetic introgression does not break down divergence (in fact, gene transfer in bacteria actually can maintain divergent genotypes--but that's a whole series of posts). Migration is really an ecological phenomenon, not one dealing with changes in genotypes. That's what I meant by "shortcomings": there are assumptions made by biologists who work on multicellular organisms (and who are the dominant force in developing evolutionary theory--why is this would, again, require a whole series of posts) that simply do not pertain to many microorganisms.
thanks