This is the second key prediction of the Big Bang: the Universe was, before any stars formed, made up of about 75% Hydrogen and 25% Helium, and much less than 1% of all other elements combined. How does the Big Bang predict this, and how to we observe it?
Well, remember we said the Universe was hot and dense in the past. At some point, it was so hot that neutral atoms couldn't form, because high energy photons would just come in and kick electrons off of nuclei. Like this:
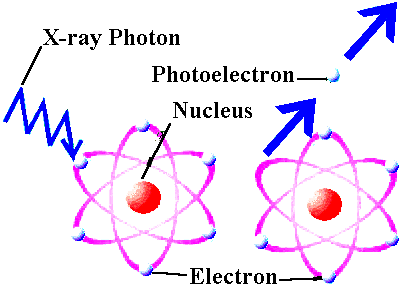
Well, you know what? At some point, even before that, the Universe was hotter and denser, and you couldn't even form stable nuclei, because things would bounce around with so much energy that they would dissociate even the smallest bound nuclei, like deuterium and helium. Well, when the Universe is hot enough, there are equal numbers of protons and neutrons. Then it cools down, and some of the neutrons decay radioactively, because neutrons are unstable. (That's right, and that's crazy. You take a typical proton, and it lives for at least 1034 years! But you take a typical neutron, and it's gone within 15 minutes.)
So you work through the math, and when the Universe finally cools down enough that you can form stable deuterium (one proton + one neutron = one deuteron), there's only about one neutron for every seven protons (the exact number depends on how many photons there are). Then all the neutrons start to fuse together, and go through reactions like this:

Well, let's do the math. I need two neutrons and two protons to make one helium nucleus, everything else becomes hydrogen. So I have two neutrons for every fourteen protons, or I'll wind up with one helium atom for every twelve hydrogen atoms.
Wait. Didn't I say 75% hydrogen and 25% helium? Yes, yes I did. But 1:12 is not the same as 25:75! Here's the deal: astronomers don't count by numbers: they count by mass. (I was livid when I first found out about this. No joke.) Well, one helium atom is four times as massive as a hydrogen atom, so the ratio of mass is 4:12, which is 25% to 75%! In all models of the big bang, this critical temperature, where neutral atoms form, is somewhere between three and four minutes into the Universe. Wait a little longer, and you'd have too little Helium. Do it a little faster, and you'd have too much. But the amount we have matches exactly what we predict.
People used to argue that it could've all started out as hydrogen, and you can make as much helium as you want in stars. That part is true. But you also make a lot of heavier elements, like Carbon, Oxygen, and Nitrogen, and sometimes Silicon, Neon, and sometimes even Iron, Cobalt, and Nickel. (And when you get a supernova, you can make anything all the way up to Uranium!) When we look at primordial gas clouds, we see less than 1% of everything besides hydrogen and helium, so that is a real tough challenge for theories that aren't the Big Bang; I haven't found one that works satisfactorily. Come back tomorrow for the last piece of the puzzle: the Cosmic Microwave Background!
- Log in to post comments





i hate you