Welcome back to The Greatest Story Ever Told, where we're bringing you the story of the Universe. We're going to go from the very beginning -- before the big bang -- up through the present day, and tell you how we got here. This is part seven, and you can always go back for parts 1, 2, 3, 4, 5, and 6. Last time, we got rid of all the antimatter in the Universe by letting it cool enough that every particle-antiparticle pair in the Universe annihilated with one another, producing a huge excess of radiation.
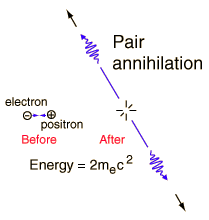
But there was also a little bit of normal matter -- protons, neutrons, and electrons -- that got left over. When we ask "how much", we found that for every ten billion photons (particles of light), we have only six protons and neutrons left over, with exactly as many electrons as there are protons to keep the Universe electrically neutral.
The Universe is also still very hot and dense, although it has been expanding and cooling. When we ended the last part, the Universe was around one second old, which means the temperature was only about a thousand times as hot as the center of the Sun. (And that's right; it took the first six parts of this story just to cover the first second!) So what do you think these protons and neutrons are going to do at these high temperatures and high densities?
Did you guess "fuse to form heavier elements" like they do in the center of the Sun? It's a good guess. After all, you certainly have enough protons and neutrons, enough energy, and a high enough density to make heavier elements. But there's a problem. For every six nucleons (protons and neutrons) that you have, you have ten billion photons. So even though you can fuse a proton and a neutron into deuterium -- the first step in our staircase towards the heavier elements -- a whole slew of photons will run into your newly formed deuterium before anything else can happen, effectively blasting it apart immediately.

If we want to make anything that lasts, we need the Universe to cool down so that these photons won't have enough energy to blast our nuclei apart. The protons will be fine; they're patient, and they live forever, so they can wait for the Universe to cool. But the neutrons? They're not so patient.
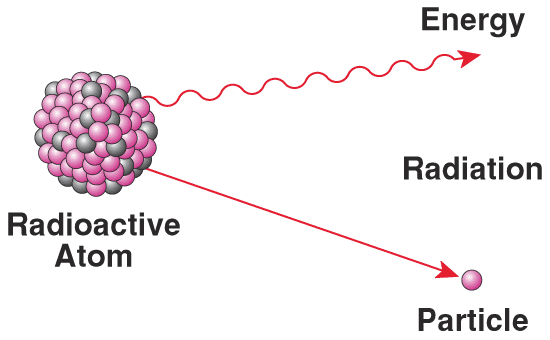
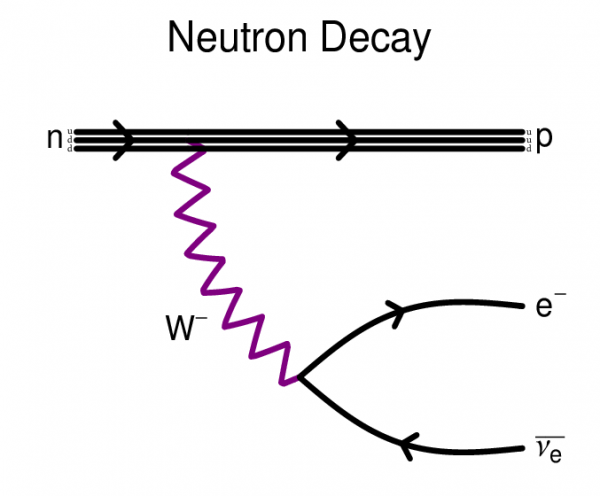
You've all heard of radioactive decay before. Sometimes, you get a collection of protons and neutrons together in a way that's unstable, and they radiate some energy away, becoming a different atomic nucleus in the process. Believe it or not, free neutrons are radioactive, too! If you leave a neutron on its own, after about ten minutes (its half-life), it will decay into a proton, an electron, and an anti-neutrino. So for every second that we wait for the Universe to cool down, so that these photons will stop blasting our nuclei apart, we lose neutrons, and replace them with protons and electrons.
If the neutrons have to wait too long for the Universe to cool, then they'll all decay, and our Universe will be 100% protons and electrons, with no neutrons at all. But our Universe is an impatient place, and when it expands, it cools fast. Every time the Universe doubles in size, the temperature halves.
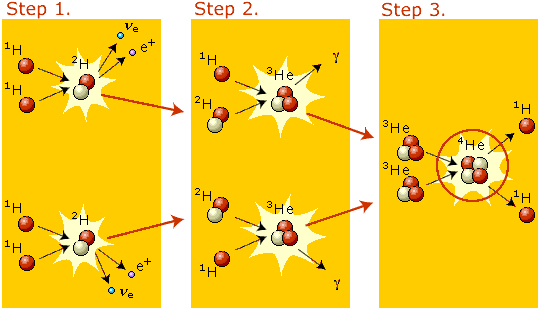
But what's really been changing quickly? The density of the Universe. Every time the Universe doubles in size, the matter density drops by a factor of eight, and the energy density (because things are also cooling) drops by a factor of sixteen. So by time the Universe is a little more than three minutes old, you can form deuterium without it being blasted apart! And once you make deuterium, everything else goes super quickly, bringing you to Helium-3, and then all the way up to Helium-4.
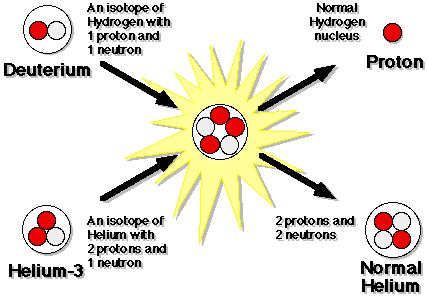
So, if you work out the math, theoretically, you find that this story should get you a Universe that was about 76% Hydrogen and 24% Helium-4, with trace amounts of Deuterium, Helium-3, and Lithium-7 from the Big Bang.
So what do you do, if you're an astronomer? You go and look for regions of space that haven't been touched by star formation since the big bang, and you try to measure how much of each element you have!
This is the earliest test we're able to perform on the Big Bang Theory: you know what elements you ought to get from the Big Bang, but what do you actually see when you go out and measure these elements? What do you find for Hydrogen, Helium-4, Deuterium, Helium-3, and Lithium-7? Let's have a look.
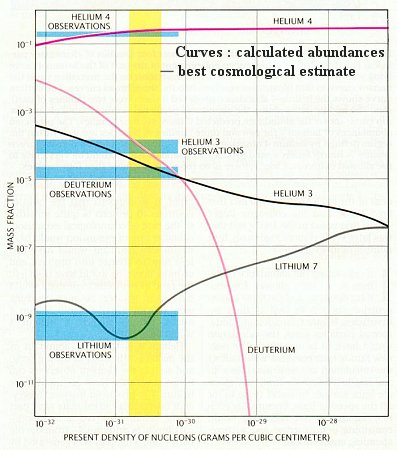
Amazingly, we measure almost exactly what we predict! This was a huge confirmation of the Big Bang Theory, and is still one of the best tools we have today!
So the Universe manages to save the neutrons by locking them up inside of -- mostly -- Helium nuclei, which are stable! We've been telling you a story that's been almost entirely theoretical, because we haven't been able to observe anything this early, until now. So our first chance to test whether this "Greatest Story Ever Told" matches reality or not is a smashing success! Not only that, but our Universe is only three minutes old at this point! What comes next? Come back for part eight...


Wow. This is a very nice work of science, a clear and concise explanation. I did not know.
But they are in a high energy environment and you just stated that a proton and a neutron fuse into deuterium and then they get blasted apart again by photons. They aren't left alone for 10 minutes, are they?
I love this series. Each time I see that there is a new exciting chapter in the story, I go and pop up a bag of popcorn and open a soda to consume while reading. A re-enactment of the old movie serial feature.
(I sure that there is an allegory between a popcorn kernel popping and the Big Bang but I'm not going to go there.)
I might have asked this question earlier, apologies if so. If we have a heap of photons after matter-anti-matter anihilate, why can't some of those photons become matter (e = mc^2)? Or is it only matter that can become energy, and not vice-versa. Or am I misunderstanding something about photons? I think of photons as weightless or just energy. After all, they travel at the speed of light and anything that had mass would require infinite energy to do that wouldn't it?
There are far too many things to learn and not enough time, but this series has helped even the balance a little. Thanks bunches!
wait! i can guess what happens next:
the universe enters puberty and asks to borrow the car.
p.s. i love the series. thank you!
@NewEnglandBob: The binding of the neutrons is a statistical process, obviously. Some neutrons get bound with a proton quickly, some others take some more time, and again some others don't manage at all to get bound before they decay. Additionally, the neutron decay is also statistical: the half-life is ten minutes means that after ten minutes half of the free ones have decayed - but even at earlier times, e. g. already in the first minute, a fair amount of them will have decayed (about 7%).
@Brian: a single photon can't "become matter" - due to conservation of momentum. But if two photons (with enough energy!) collide, indeed matter particles can be formed. At that indeed happened all the time in the earliest times of the universe (as far as I know, approximately in the first second - afterwards the photons had not enough energy).
So when Ethan says:
we got rid of all the antimatter in the Universe by letting it cool enough that every particle-antiparticle pair in the Universe annihilated with one another, producing a huge excess of radiation.
A lot of the matter found in the universe could have been created in that first second then?
But there was also a little bit of normal matter -- protons, neutrons, and electrons -- that got left over. When we ask "how much", we found that for every ten billion photons (particles of light), we have only six protons and neutrons left over, with exactly as many electrons as there are protons to keep the Universe electrically neutral. Was that all left over, or did some or all of it come from photons? Seems to me that with conservation of energy, it would make more sense that all matter came from photon collisions than left over by chance.
Maybe I've got this completely wrong, but wouldn't photon-photon collisions produce matter/antimatter pairs, not simply matter particles? I think we're running in to an ambiguity of wording between matter as distinct from energy and matter as opposed to antimatter.
@Brian: at these earliest times, matter (or more precisely, as Morgan pointed out, matter-antimatter pairs) is created all the time from photon collisions (and gluon collisions etc.) - but also the matter and antimatter particles annihilate each other all the time. There was a very hot "soup" of photons, electrons, positrons, quarks, gluons, etc., which all reacted with each other all the time. So it's hard to state precisely what matter "came from photons" and what was "left over" - essentially, all matter was both at the same time.
So it's hard to state precisely what matter "came from photons" and what was "left over" - essentially, all matter was both at the same time.
Cool. Thanks. I'm really ignorant about this stuff. Been years since I finish high-school physics and am only just starting to get back into it so I'm sure my questions probably are not even wrong.
So both e = mc^2 and e = ~mc^2? That is, energy is both matter and anti-matter? :)
The "m" there is mass, not "matter". Matter has mass, and antimatter has mass, and they're the same kind of mass (unlike hypothetical exotic "negative mass" stuff you hear about when talk turns to wormholes and warp drives and so on).
The "m" there is mass, not "matter". Matter has mass, and antimatter has mass, and they're the same kind of mass
So antimatter is just matter? (Matter has mass, something you can weigh or is affected by gravity or resists change in momentum and likewise antimatter.) I thought antimatter was some exotic opposite of matter that mutually destructed with contact with its opposite. If a proton is positive and an antiproton is negative in charge (like an electron, only bigger). why is an antiproton exotic? Why does it anihilate a proton if they're both just mass? Quite confused now.
@Brian: Anti-matter is in a sense the "opposite" of matter (it has the opposite electric charge, and some other things like "baryon number" also have the opposite sign) - but the only sense in which it could be called "exotic" is that there is so little anti-matter in the universe, compared to the amount of matter (see Ethan's part 6 for details why this is so). In all other respects, it behaves essentially exactly like "normal" matter.
Oh, and an antiproton is *not* simply "like an electron, only bigger". An antiproton consists of three anti-quarks (plus gluons plus see-quarks), whereas an electron is, as far as we know, an elementary particle. And an antiproton can annihilate with a proton to photons (or other particles) because that reaction does not violate any conservation laws - that's all.
Bjoern
#8 you said, "a single photon can't "become matter" - due to conservation of momentum. But if two photons (with enough energy!) collide, indeed matter particles can be formed."
Let me pick nits and clarify (or perhaps I'm confused). Photons exist only if there are also electrically charged particles (e.g. quarks and leptons). There is no such theoretical QED object as a "single isolated photon" or "single isolated electron". Furthermore, two photons can only "become matter" if two "electrically charged particles" already exists (one electrically charged particle is not enough), (I can probably find a Feynman quote).
In other words, the existence of an electromagnetic field (i.e. photons) already implies the existence of electromagnetic particles (i.e. particle and antiparticle leptons or quarks). Thus the "fundamental electromagnetic entity" is not a "single photon" or "single electron" (each of which is a non sequitur); the fundamental electromagnetic entity is a collective open system of indeterminant number of particles (i.e. of photons and electrically charged fermions). Thus Ethan's FIRST DIAGRAM (above) is technically incorrect; his BEFORE picture need photons and his AFTER picture needs particles and antiparticles.
Now my point in explaining this (is not to support the big bang which is really irrelevant to this phenomenon) but to emphasize that there is an equilibrium BETWEEN fermions (e.g. electrons and positrons) and bosons (photons) that is TEMPERATURE DEPENDENT. It's similar to a chemical reaction, e.g. raise the temperature you get more H2 and O2, lower the temperature you get more H2O.
Which brings us back to the one of physics biggest unresolved questions: where are all the missing antiparticles?
NOW, THE THING THAT interests me the most in this post is Ethan's LAST DIAGRAM. I was hoping someone might discuss my question, without me asking it.
Here's my question. How TEMPERATURE DEPENDENT is the theoretical calculations of Helium 4, Helium-3, Deuterium and Lithium abundances? Specfically, "If the neutrons (DON"T)have to wait too long for the Universe to cool" because there wasn't a big bang; then would the protons and neutrons still just fuse (with the same relative abundance of He-4, He-3, De, Li) in our "steady state" 2.7K universe. Thus starting with the same starting distribution of protons and neutrons (as in the big bang theory), but with a different temperature history (steady state 2.7K) what is the relative distribution? Thanks.
@Thomas:
Err, yes, there is. I don't know why you think otherwise...? A universe with only a single (real) photon, but no (real) electrons or other charged particles, in it would be a valid solution of the equations of QED.
You only need virtual electrically charged particles, not real ones.
Better than an argument from authority: open up a book on QED and do the work yourself. But if you don't know how to do that, a quote would be nice...
Actually, it's the other way round: the local U(1)-gauge symmetry of particles like the electron implies the existence of an electromagnetic field. But from that it doesn't follow that a photon or an electron couldn't also exist on its own - field and particles are not identical. You can have a field without any (real) particles.
The reaction is temperature dependent - but even more important: the reaction rate obviously depends on the density! With the density we have today, there is no way at all to get almost all of the neutrons fused before they can decay!
Even the most ardent supporters of steady-state models (Hoyle, Narlikar et al.) admit that the universe *must* have been in a much denser, much hotter state at earlier times - because you can't get the observed elements (and the CMBR, etc.) in any other way.
The physical properties of deuterium compounds can exhibit significant kinetic isotope effects and other physical and chemical property differences from the hydrogen analogs; for example, D2O is more viscous than H2O.[7] Chemically, deuterium behaves similarly to ordinary hydrogen, but there are differences in bond energy and length for compounds of heavy hydrogen isotopes which are larger than the isotopic differences in any other element. Bonds involving deuterium and tritium are somewhat stronger than the corresponding bonds in hydrogen, and these differences are enough to make significant changes in biological reactions
Great work Ethan!You have explained the universe story so well.I was unaware of certain facts.It was refreshing.Thanks
As a social scientist, I had never got myself to go past the first page of books on the big bang and black holes. This post is written with such passion that I found myself reading it to the end. The post reminds me of Stephen Hawkins' works in the field. I will be tearing down my basement in search of his works that I had, for years thought were too technical for me. That he's had to surmount incredible physical challenges in his daily and professional life will themselves be inspiration enough to read on. Kudos Ethan!
Incredible! Which authoritative books can a non-techie like me get hold to learn more about the fascinating phenomena mentioned in this page?
Amazing! I've always been curious about neutrons & the big bang theory. Hopefully this will prove exactly how our universe got started!
This series has been a lot of fun to read. Funny story.. My two uncles were having a heated debate.. one a creationist, the other science teacher.. The creationist stated that the Big Bang Theory is the most absurd thing he'd ever heard of.. "everything all of a suddent came from nothing, now come on" he exclaimed.. The science teacher looked at him and said.. "Well, isn't that exactly what you believe?"
-Peter, grout stain guy
There are far too many things to learn and not enough time, but this series has helped even the balance a little. Thanks allot!
Frank- Columbus Ohio Auto Insurance Quotes
At the level of theory, they're wrong: supersymmetry is much more beautiful and nontrivial a symmetry than some simple past-future reversal. And it's even plausible that they will be proved wrong experimentally in the near future, too.
Annuities
I've always been curious about neutrons & the big bang theory as well as other different theories. Some furniture companies also follow some of the theories in there collection like castelle patio furniture.
Actually, it's the other way round: the local U(1)-gauge symmetry of particles like the electron implies the existence of an electromagnetic field. But from that it doesn't follow that a photon or an electron couldn't also exist on its own - field and particles are not identical. You can have a field without any (real) particles.
xbox live code generator
The big bang starts off with a state of extremely high density and pressure for the Universe. Under those conditions, the Universe is dominated by radiation. The majority of the energy is in the form of photons and particles (like neutrinos) that move at near the speed of light. As the big bang evolves in time, the temperature drops rapidly as the Universe expands and the average velocity of particles decreases.
Gift Ideas for Men
When I took my natural sciences course at the uni, there was the same theme about the Universe, but then I found it too boring to get into the details. However, this post is really very interesting, and I discovered some new stuff for me I had no idea about before.
It doesn't look like anything I've learned about the Universe at school. Thanks for descriptive explanation and nice illustrations.
The doctors' lobby and the hospitals' lobby are also on board with the Senate bill.
Does 'Wall-E' really single the people out as Americans? If not, I'm pretty sure it is an interpretation -- and, yes, "sub"text -- to infer that they are meant to only represent Americans. I haven't seen the movie, but reading that description, it sounds as if the point is that the entire human race has evolved into perfect consumers, which would perfectly fit with what I have been led to believe the theme is.
The big bang starts off with a state of extremely high density and pressure for the Universe. Under those conditions, the Universe is dominated by radiation. The majority of the energy is in the form of photons and particles (like neutrinos) that move at near the speed of light. tv episodes guide
And the President reflects on that. This is a President who is really reflecting on his place in history."
It was awesome to meet the entire RFGO crew, you guys are amazing!
This is part seven, and you can always go back for parts 1, 2, 3, 4, 5, and 6. Last time, we got rid of all the antimatter in the Universe by letting it cool enough that every particle-antiparticle pair in the Universe annihilated with one another, producing a huge excess of radiation
Well it's just a theory that tries to explain it but like all others it just doesn't cut it. It apparently cannot explain what caused the existence or creation of the very first universes that are always existing in this cyclical evolution of each universe. It does not explain what, why and how the very first universe came into being and how time arise. It will take us much longer to get behind this..
Really interesting! I really like this series. Although its a bit confusing & I'd have to read it a few times, I actually feel a bit smarter now.
Great approach!! congratulation man!, is amazing how you achive this by using very simple tools. Man you are a god. When I grow up, I wanna be like you.
So the big bang starts in a state of really high density and pressure? Is there any books out there you can recommend that simplifies this so that someone such as myself can understand better? Thanks
My ignorant understand is that the radioactive waste from these fusion reactions generally has a much shorter half life than those of fission.
Submited by : Dietas Peso
We use it all and then we move on, no idea what the consequences are. But who cares where our children have to live, we lived good.
Foreclosure Process in Alaska
For example, many people cannot afford medicines for these or other diseases, even though some are easily treatable.
lasik surgery
great post , really good perspective on the subject and very well written, this certainly has put a spin on my day, many thanks from the UK and keep up the good work
Interesting read, was not what I was expecting but that's the great thing about this website, keep up the good work
you can recommend that simplifies this so that someone such as myself can understand better. It will take us much longer to get behind this.
in a state of really high density and pressure? Is there any books out there you can recommend that simplifies this so that someone such as myself can understand better?
If not, I'm pretty sure it is an interpretation -- and, yes, "sub"text -- to infer that they are meant to only represent Americans.training for can
Incredible! Which authoritative books can a non-techie like me get hold to learn more about the fascinating phenomena mentioned in this page?
So the big bang starts in a state of really high density and pressure? Is there any books out there you can recommend that simplifies this so that someone such as myself can understand better? Thanks
The universe is '76% Hydrogen'? I dont deny it but how come it doesnt explode?
Was helping my daughter tonight with her homework on the Big Bang and this article was very helpful, thanks
so amazing I can't imagine why they can do that and how can they take this activities ?
Thanks for providing very interesting details about the universe. It is very interesting to hear that there are protons and electrons in the universe.
We use it all and then we move on, no idea what the consequences are. But who cares where our children have to live, we lived good.
This article is a clear indicator that I have forgotten most of the 4 physics classes I took many years ago. Thanks for the refresher course. It was a good start.
There is no such theoretical QED object as a "single isolated photon" or "single isolated electron".
"I agree that ABC is better than XYZ, especially when it comes to quality. I have been using ABC for the last 3 years it never dissapointed me. Does anybody know when the new version is coming out?"
Hopefully, with the experiments continuing to happen at CERN we can learn more about the evolution of the universe and the composition of matter within it.
He also described how he once came across an interesting-looking alternative to the Big Bang. However, he discovered that the numbers did not work out. Millimeter-sized black holes would produce Hawking radiation with the right temperature, but the necessary mass density of them would be too high.
Well it's just a theory that tries to explain it but like all others it just doesn't cut it. It apparently cannot explain what caused the existence or creation of the very first universes that are always existing in this cyclical evolution of each universe. It does not explain what, why and how the very first universe came into being and how time arise. It will take us much longer to get behind this..
He also described how he once came across an interesting-looking alternative to the Big Bang. However, he discovered that the numbers did not work out. Millimeter-sized black holes would produce Hawking radiation with the right temperature, but the necessary mass density of them would be too high.
The density of the Universe. Every time the Universe doubles in size, the matter density drops by a factor of eight, and the energy density (because things are also cooling) drops by a factor of sixteen. So by time the Universe is a little more than three minutes old, you can form deuterium without it being blasted apart!
After all, you certainly have enough protons and neutrons, enough energy, and a high enough density to make heavier elements. But there's a problem. For every six nucleons (protons and neutrons) that you have, you have ten billion photons. So even though you can fuse a proton and a neutron into deuterium -- the first step in our staircase towards the heavier elements
The density of the Universe. Every time the Universe doubles in size, the matter density drops by a factor of eight, and the energy density (because things are also cooling) drops by a factor of sixteen. So by time the Universe is a little more than three minutes old, you can form deuterium without it being blasted apart! And once you make deuterium, everything else goes super quickly,
My chemistry is week somehow. But I loved your srticle and understand it through pictures.
deuterium behaves similarly to ordinary hydrogen, but there are differences in bond energy and length for compounds of heavy hydrogen isotopes which are larger than the isotopic differences in any other element.
The density of the Universe. Every time the Universe doubles in size, the matter density drops by a factor of eight, and the energy density (because things are also cooling) drops by a factor of sixteen. So by time the Universe is a little more than three minutes old, you can form deuterium without it being blasted apart! And once you make deuterium, everything else goes super quickly, bringing you to Helium-3, and then all the way up to Helium-4.
Well it's just a theory that tries to explain it but like all others it just doesn't cut it. It apparently cannot explain what caused the existence or creation of the very first universes that are always existing in this cyclical evolution of each universe. It does not explain what, why and how the very first universe came into being and how time arise. It will take us much longer to get behind this..
Well it's just a theory that tries to explain it but like all others it just doesn't cut it. It apparently cannot explain what caused the existence or creation of the very first universes that are always existing in this cyclical evolution of each universe. It does not explain what, why and how the very first universe came into being and how time arise. It will take us much longer to get behind this..
eutrons are constituted by 1 up quark and 2 down quark
up quark has a charge of +2/3e
down quark has a charge of -1/3e
the total sum of charges in a neutron is 0, no charge.
This is part seven, and you can always go back for parts 1, 2, 3, 4, 5, and 6. Last time, we got rid of all the antimatter in the Universe by letting it cool enough that every particle-antiparticle pair in the Universe annihilated with one another, producing a huge excess of radiation
The density of the Universe. Every time the Universe doubles in size, the matter density drops by a factor of eight, and the energy density (because things are also cooling) drops by a factor of sixteen. So by time the Universe is a little more than three minutes old, you can form deuterium without it being blasted apart! And once you make deuterium, everything else goes super quickly, bringing you to Helium-3, and then all the way up to Helium-4.
The density of the Universe. Every time the Universe doubles in size, the matter density drops by a factor of eight, and the energy density (because things are also cooling) drops by a factor of sixteen. So by time the Universe is a little more than three minutes old, you can form deuterium without it being blasted apart! And once you make deuterium, everything else goes super quickly, bringing you to Helium-3, and then all the way up to Helium-4.
With the right program, anyone can see what a computer is doing wirelessly on the internet. If you will be embarrassed if someone knew you were watching porn online then you are too immature for it. Also, if the hotel offers FREE wifi then why are you asking if you will see something on your bill? The TV is free to watch, will you get an itemized bill showing every time you turned on the TV? No. So don't be so naive.
Hi, I'm Politiko. I'm here simply as a political observer to chime in with the likes of Messrs. Gatemouth, Hackshaw, Yoda, Enwhyseawonk, Gumbs and Littlefield (where, by the way, are the women? Ms. Markowitz seldom posts and might not really be female)
I will be tearing down my basement in search of his works that I had, for years thought were too technical for me.
Well it's just a theory that tries to explain it but like all others it just doesn't cut it. It apparently cannot explain what caused the existence or creation of the very first universes that are always existing in this cyclical evolution of each universe. It does not explain what, why and how the very first universe came into being and how time arise. It will take us much longer to get behind this..
While my wife was teaching animation at the Art Institute of Atlanta, she half-kiddingly proposed a new major... Dermographics, the art, science and otherwise of Tattoo. Recognizing that one couldn't teach comic book art in college without renaming it Sequential Art, because what parent would fork over the dough or proudly brag to friends that their precious spawn was pursuing a degree in Comic Books...
Dermographics. The new "Plastics"...
Wait for it. It will happen.
daytrading
Uhm maybe I've got this completely wrong, but wouldn't photon-photon collisions produce matter/antimatter pairs, not simply matter particles?
I am sure we're running in to an ambiguity of wording between matter as distinct from energy and matter as opposed to antimatter right?
The density of the Universe. Every time the Universe doubles in size, the matter density drops by a factor of eight, and the energy density (because things are also cooling) drops by a factor of sixteen. So by time the Universe is a little more than three minutes old, you can form deuterium without it being blasted apart!
May I print and use this for my science class? I try (and sometimes successfully) teach under 10s the benefits of understanding the universe from the smallest parts upwards. Otherwise its too difficult to comprehend, like trying to teach a fully grown adult how to optimize an Annuity.
This totally crushed my skull. I'll give like $5 to someone who can put this in laymens terms!
I find it very interesting how actual physics and the physics used in Star Trek are quite similar. Not to mention it appears that all the nebula's that were shown look as though they are real. I cant believe how close the antimatter is to Star Trek.
Are there any long term solutions being proposed or are we turning a blind eye on the situation and letting the next generation worry about it? www.itunes.com
Excellent post, one of the few articles Iâve read today that said something unique! One new subscriber here :)
Excellent post, one of the few articles Iâve read today that said something unique! One new subscriber here :)
Among the greatest story ever said and i would love to read it again and again. Now in the modern society, radioactive element plays the key important role to develop top class material and high power capacity of electricity.
Your post is terrific and really insuperable.
Excellent post, one of the few articles Iâve read today that said something unique! One new subscriber here :)
Well it's just a theory that tries to explain it but like all others it just doesn't cut it. It apparently cannot explain what caused the existence or creation of the very first universes that are always existing in this cyclical evolution of each universe. It does not explain what, why and how the very first universe came into being and how time arise. It will take us much longer to get behind this..
Does 'Wall-E' really single the people out as Americans? If not, I'm pretty sure it is an interpretation -- and, yes, "sub"text -- to infer that they are meant to only represent Americans. I haven't seen the movie, but reading that description, it sounds as if the point is that the entire human race has evolved into perfect consumers, which would perfectly fit with what I have been led to believe the theme is. Thanks.
Well it's just a theory that tries to explain it but like all others it just doesn't cut it. It apparently cannot explain what caused the existence or creation of the very first universes that are always existing in this cyclical evolution of each universe. It does not explain what, why and how the very first universe came into being and how time arise. It will take us much longer to get behind this..
He also described how he once came across an interesting-looking alternative to the Big Bang. However, he discovered that the numbers did not work out. Millimeter-sized black holes would produce Hawking radiation with the right temperature, but the necessary mass density of them would be too high.
Does 'Wall-E' really single the people out as Americans? If not, I'm pretty sure it is an interpretation -- and, yes, "sub"text -- to infer that they are meant to only represent Americans. I haven't seen the movie, but reading that description, it sounds as if the point is that the entire human race has evolved into perfect consumers, which would perfectly fit with what I have been led to believe the theme is.
Well it's just a theory that tries to explain it but like all others it just doesn't cut it. It apparently cannot explain what caused the existence or creation of the very first universes that are always existing in this cyclical evolution of each universe. It does not explain what, why and how the very first universe came into being and how time arise. It will take us much longer to get behind this..
The "m" there is mass, not "matter". Matter has mass, and antimatter has mass, and they're the same kind of mass (unlike hypothetical exotic "negative mass" stuff you hear about when talk turns to wormholes and warp drives and so on).
I haven't seen the movie, but reading that description, it sounds as if the point is that the entire human race has evolved into perfect consumers, which would perfectly fit with what I have been led to believe the theme is. Thanks.
Excellent post, one of the few articles Iâve read today that said something unique! One new subscriber here. à¸à¸³à¸à¸±à¸à¸à¸¥à¸§à¸
If the point is that the entire human race has evolved into perfect consumers, which would perfectly fit with what I have been led to believe the theme is. Thanks.
There are plenty more things to learn but haven't not enough time. This series has helped me a lot. Thanks for sharing!
I love this series. Each time I see if there is a new exciting chapter in the story. Keep up the posting, thanks.
I haven't seen the movie, but reading that description, it sounds as if the point is that the entire human race has evolved into perfect consumers, which would perfectly fit with what I have been led to believe the theme is. Thanks.
Nice pics and great explanation on this topic so debated for thousands of years.
Great story + awesome photos = super article
so, save the neutrons and you'll save the world!
This is a great story. Thank for sharing with us. You did a great job on this mate!
This is a great story. Thank for sharing with us. You did a great job on this mate! Locum Tenens Physician
Fascinating theme this one with the neutrons and the world.
so, this is how Mother Tera was born...
So - step1 - step2 - step3 and here we are! :)
Right, so if the universe started out as a few elements in a hot dense state then what created that hot dense proto-universe? How did it come to be? Surely it didn't just appear out of nothingness did it?
Its always good to have tips on good blog posting. As I just started posting comments for blog and faced a lot of rejections. I think your suggestion would be helpful for me. I will let you know if this works for me.
Excellent post, really good perspective on the subject and very well written, this certainly has put a spin on my day, many thanks from UK and keep up good work.
now that we know the "recipe" let's make another Tera...
Interesting what science geeks consider to be a good story!
We have enough gun control. What we need is idiot control.
Well it's just a theory that tries to explain it but like all others it just doesn't cut it. It apparently cannot explain what caused the existence or creation of the very first universes that are always existing in this cyclical evolution of each universe. It does not explain what, why and how the very first universe came into being and how time arise. It will take us much longer to get behind this..
Hi, I am in search of a good shared hosting company. Could you suggest one to me? I could sign up by using an aff link you give me. My current hosting firm is pretty shaky right now. :-/
It apparently cannot explain what caused the existence or creation of the very first universes that are always existing in this cyclical evolution of each universe. It does not explain what, why and how the very first universe came into being and how time arise. It will take us much longer to get behind this..
There are far too many things to learn and not enough time, but this series has helped even the balance a little. Thanks allot!
There are far too many things to learn and not enough time, but this series has helped even the balance a little. Thanks allot!
Ethan - may I ask a question about these statements in your presentation? "This is the earliest test we're able to perform on the Big Bang Theory. Amazingly, we measure almost exactly what we predict! This was a huge confirmation of the Big Bang Theory." Exactly who conducted those measurements, using what equipment, when? Can you point me to an article, study or website for some detail? I love this stuff, but like to know the sources of hard validation so I can be comfortable knowing that I'm loving more than my blind faith. Thanks, I love your work and your ability to communicate it so clearly.
bwiley,
You can find a review of the status of cosmological big bang nucleosynthesis in the Particle Data Group at Lawrence Berkeley Labs' review of particle physics, published biannually.
Here is a link to the most recent update I can find, 2006, as of this time.
Ethan,
Many thanks, that was a good place to start. It seemed that the confirmation would have been such a landmark occurrence that I was wondering why it wasn't more prominent in the literature. Ultimately, the document you referred me to lead me to this one, which I found the most concise for my own curiosity's purposes: Review of Big Bang Nucleosynthesis and Primordial Abundances; http://xxx.lanl.gov/abs/astro-ph/0001318
I provide it just in case you or anyone that might read this far down into the comments is curious. Anyway, thanks again, and I love reading your work. Really, it's some of the best I've come across.
Was helping my daughter tonight with her homework on the Big Bang and this article was very helpful, thanks
We have enough gun control. What we need is idiot control.
I haven't seen the movie, but reading that description, it sounds as if the point is that the entire human race has evolved into perfect consumers, which would perfectly fit with what I have been led to believe the theme is. Thanks.
I find it very interesting how actual physics and the physics used in Star Trek are quite similar. Not to mention it appears that all the nebula's that were shown look as though they are real.