Why must I feel like that?
Why must I chase the cat?
Nothin' but the dog in me. -George Clinton
(And I'll get it stuck in your head, too, if you like. Have a go at the extended 10-minute version.)

In other words, it might not be as catastrophic as it once was, but the Universe is still just a super hot, dense plasma filled with a huge amount of radiation. Well, what happens now? The surprising answer -- for thousands of years -- is nothing interesting.
Why not? Well, we've got a whole Universe full of heavy, positively charged nuclei and light, negatively charged electrons. The Universe is too cold to blast the nuclei apart, but too hot to form neutral atoms!
I mean, you can form them for an instant, sure, but remember that for every atomic nucleus, you have over a billion photons. If even one of them has enough energy, it's going to blast that electron not just to a higher energy level (as shown below), but right out of the atom!

So, if we want to do anything else with our Universe, we first need to make some neutral atoms. And the only way to do that? We've got to wait. Our Universe needs to expand and cool enough for this to be possible, and unlike nucleosynthesis, where we had to wait a few minutes, we have to wait hundreds of thousands of years!
Why do we have to wait so long? Take a look at the energy spectrum of our photons -- the particles of light in the Universe -- which was made famous by xkcd.
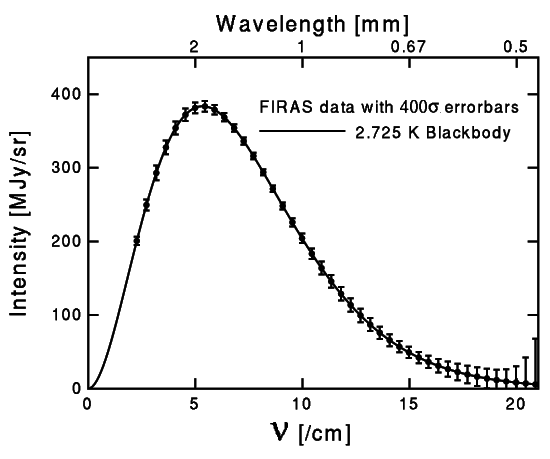
It takes 13.6 eV of energy to ionize a hydrogen atom. But that doesn't mean that the average energy of the Universe needs to drop below 13.6 eV. Look at that graph above. Notice how, even though the average photon has a frequency of around 6 in the graph, there are still plenty with a higher frequency (and hence higher energy). In fact, because photons are so numerous (again, outnumbering nuclei by a factor in the billions), we need the average energy to drop down to about 2% of the ionizing energy!
Well, the only way the energy drops is because the Universe cools as it expands. Light -- all light -- has a wavelength, and the longer the wavelength, the lower the energy, and hence the cooler it gets. As the Universe expands, the wavelength gets stretched out, and so the Universe gets colder!

So now the question becomes: how long do we have to wait for the Universe to cool down this much? And sadly, the answer is 380,000 years! It took 7 parts to just tell some highlights from the first five minutes of the Universe, and then almost nothing of interest happens for hundreds of thousands of years! No stars, no galaxies, nothing. We have to wait this long just to get neutral atoms!

But once you get them, you're in business! All of a sudden, the light that used to bounce off of protons, electrons, and other nuclei now simply travels in a straight line! All of those billions and billions of photons, once we form neutral atoms, don't interact with anything anymore.
Until, that is, they run into something. Like, for instance, us. Can we see this leftover radiation from the big bang?

You bet we can! In the 1960s, we discovered the leftover radiation from the big bang, which has now shifted into the microwave! In fact, in the 2000s, we measured the fluctuations in the microwave background, and what did we find?

The fluctuations are exactly the kind we expect to get from inflation! These observations are huge verifications of our picture of how the Universe work. In fact, just this -- the story of the formation of neutral atoms and the leftover glow -- was to lead to the widespread acceptance of the big bang and the rejection of pretty much every alternative. Why? Because they don't predict this leftover glow that we see.
So -- for those of you who don't like the big bang -- this is your number one challenge when you make a theory of the Universe. Can you explain the observed abundances of the elements? Can you explain the leftover microwave radiation? Can you explain the fluctuations in that microwave radiation? And can you explain the expansion of the Universe?
These first eight parts have put us in a great place, and we're now set up -- with a Universe full of neutral atoms -- for things to get both interesting and familiar! See you next time...
- Log in to post comments

Ethan,
I'm enjoying your series, thanks for your effort.
Could you explain the following; given the conservation of energy and the photons losing energy as the universe expands, how do the photons shed the energy and where does it go? Of course the real question is, what am I missing?
But wouldn't it be possible for gravity to already start to form denser areas? After all, density works just fine on plasma. Doesn't that count as something happening?
Thanks for the Atomic Dog. It made me realize I forgot that one in my iTunes library.
Not only are you an astrophysics freak, you're funkdafied.
That said I've enjoyed this series. This one in particular with the CBR discovered by Penzias and Wilson of Bell Labs is a particular "In your face" for those who don't accept the Big Bang theory.
In the list of links to previous installments you've linked to part 7 twice and not to part 6 :P
Loving the series, by the way, I never knew how sophisticated and interesting a theory the old "Big Bang" really is.
I am really enjoying this explanation of the big bang. Thank you!!
The Universe has also expanded and cooled off a lot during these first few minutes;
The only "observers" at this time would be the particles, moving very very fast, so according to GR, wouldn't that "few minutes" for them be a very long time, esp. if they were travelling close to c?
But wouldn't it be possible for gravity to already start to form denser areas? After all, density works just fine on plasma. Doesn't that count as something happening?
I believe what's happening at this early stage is that the e's and p's have so much energy of motion from being bashed by photons that it swamps any nascent "concentrations" from coalescing, for the same reason that at subatomic distances e's cannot even maintain stable orbits with p's to form hydrogen atoms.
Energy isn't conserved in general relativity.
Nothing to do with that, Sili. As the Universe expands, the light waves get stretched out, to lower frequencies, and a photon with a lower frequency has a lower energy. But one gamma ray photon can be converted to several X-ray photons, which can be converted to several ultraviolet photons, which can... you get the idea. :P Same thing happens in the Sun's radiative zone as photons scatter off nuclei and electrons as they make their way up to the surface.
Great! Where was I during parts 1-7 - I'll have to view them. How about putting them all on a Youtube channel where we can watch the whole thing in nice little 10-minute segments? I have watched quite a number of science lectures in that format and it's a great way to experience them. You can watch 10 minutes a day (like with breakfast or before bedtime) and run through an hour lecture in a week. Plus sleep on all the concepts while your absorbing the information. Great for educating the proles (like me!)
#9 Procyon
Well how about now? A photon emitted by an excited hydrogen atom in NGC 4351, as it travels to your eye, gets stretched to lower frequency, i.e., reddens or gradually loses energy. Where did the energy go?
Sili is correct. The answer to #1: in a universe where space itself is expanding, energy is not conserved.
Perhaps Ethan can devote a blog entry to this topic. :)
zeke
When space itself expands as a photon travels, the photon's velocity stays constant, its wavelength stretches and energy is conserved. This is the difference between normal motion and Hubble motion. This is because c is a constant. If it were not, a photon released from a star moving rapidly toward you would have a speed in excess of c. This illustrates the difference between motion within space and motion due to the expansion of space. With the latter, you are not even moving relative to things near you. With the former, you are.
Another way to view this is that the stretching of a photon's wavelength is a direct measure of the stretching of space. In a static piece of space, a photon's wavelength should stay constant. When its wavelength stretches, that is a sign that space itself is stretching. This is what Edwin Hubble discovered.
I happen to be reading Weinberg's "The First Three Minutes" at the moment. I know it's an old book, but unless I missed something he put the question in terms of energy density. The reason the temperature drops is exactly analogous to a gas being released from pressure. Fewer photons per liter of volume of space means less energy per liter of volume of space which is cooler temperature per unit volume. The overall energy on the total universe remains the same. I'm not saying any of the other posters are wrong. I honestly am way over my head here. I can't even say that this is what Weinberg was trying to convey... just that this is what I got from reading this book.
I see our host covered this in lecture #4.
What prevents the whole mess from collapsing int a black hole at this stage? We have a lot of mass in very close proximity, how come gravity isn't the dominant force?
@Douglas Watts: You are contradicting yourself. If c is constant (and probably most people would agree with that ;-) ), but the wavelength increases (redshift), the frequency decreases (since always c = wavelength times frequency!). But the energy of a photon is Planck's constant times the frequency. Hence the energy of a photon decreases - it doesn't stay constant, as you claim.
@Jamie: The overall energy of (non-relativistic) matter stays the same, the overall energy of radiation decreases (inversely proportional to the scale factor), the overall energy of "dark energy" increases (proportional to the third power of the scale factor). For the energy densities, it is: (non-relativistic) matter goes inversely to the third power of the scale factor, radiation inversely to the fourth power, and dark energy density stays constant.
Bjoern. Thanks. My mistake. Increase in wavelength = decrease in frequency = less energy. My point was that because c is a constant, this is the only way a photon can lose energy.
Why would anyone not like the Big Bang? It always has such nice things to say about everyone.
I thought inflation was proposed to explain the observed fluctuations? Doesn't this statement misrepresent the causal relationship between the theory and observation?
Also, sorry to nitpick, but shouldn't you avoid using such misleading illustrations of atoms? I think Wikipedia has plenty of illustrative pictures on either the atom article or the article on electron orbitals, which would be much more informative and help us in taking a step towards dispelling public myths.
@Cody: As far as I know, inflation was mainly proposed to explain the observed homogenity of the CMBR and the flatness of the universe, not to explain the observed fluctuations. After all, the fluctuations were only discovered by COBE, *long* after inflation was first proposed!
For those of you who like the big bang, Wiki "HE0107-5240 is an extremely metal-poor Population II star, located roughly 36,000 light years away from the Earth,... with an age of roughly 13 billion years."
Now one possible reason that the oldest star in our Milky Way galaxy is essentially of the same age as the "hypothetical age of the universe" (13.6 billion years) is that any star is either blown to bits, exhausts its fuel or is swallowed by the supermassive black hole in the center of our galaxy within the galaxy/(black hole at center of galaxy) life cycle of 13.6 billion years.
Which brings me to the point that I'd really like to better understand: how long does it takes to build a black hole of up to a billion solar masses at the center of a galaxy. If the universe is 13.6 billion years old; then a black hole at the center of a 13 billion years old galaxy (such as our Milky Way) would need to swallow 1 stars every 10 years to become a 1 billion star black hole, or 1 star every 10,000 years to become a 1 million star black hole. Remember our Milky Way has 100 to 400 billion stars.
So the astronomical question is: how many KNOWN stars at the center of galaxies disappear from view every year? Assuming that such massive black holes do occasionally swallow stars and don't always slingshot then to the other end of their galaxy. Focusing upon the stars at the centers of various galaxies (including our Milky Way), have any central stars disappeared from view in the last 10 years of intense automated astronomical observation?
I haven't seen any reports of missing stars. Rather the stability of even binary star systems suggests that the merging of stars even when the companion is a black hole is a very rare event. So show me the math, knowing the collision rate of stars in a spiral galaxy; how long does it take to form a 1 million or 1 billion solar mass black hole? I suspect that we haven't seen this calculation because the answer is considered absurd (e.g. a 100 billion or 1 trillion years or such). Hence, until the formation of supermassive center of galaxy black hole can be described (fudged) in a way that agrees with the big bang theory the answer will be under intense investigation.
Let me not repeat the many other extraordinary assumptions which cause me to remain skeptical of the big bang theory. And by the way, yes there are just as many if not more reasons to remain skeptical of any steady state model. But science is not politics. When faced with the choice between two seriously flawed theories the big bang theory (better but seriously flawed) and the various current steady state theories (worse than seriously flawed); we do not have to accept the better but seriously flawed theory. I mean is a chain of reasoning with 20 weak links (the big bang theory) really any better than a chain of reasoning with 30 weak links (the various current steady state theories).
The possible lifetime of small-mass stars is *much* longer than 13.6 billion years. And I see no way how all such stars which are created far out in a galaxy should manage to be swallowed by the Black Hole in the center in these 13.6 billion years. Hence the fact that we haven't observed any stars older than about 13 billion years is a nice confirmation of the Big Bang theory.
Here you neglect to consider several things: 1) Most matter in the universe is not in the form of stars, but gas and dust clouds; obviously we can't observe directly when such clouds are swallowed by a Black Hole. 2) The only galaxy where we have enough optical resolution to observe individual stars in the central part, close to the Black Hole, is our own Milky Way (as far as I know). And the Black Hole in the Milky Way is quite inactive in the moment; some stars orbit it, but none is close enough to be swallowed in the next few thousands (or even million?) years. 3) The growth of the Black Holes isn't constant; it was far greater in earlier times of the universe. Observational evidence: we can see far more active galaxies (especially quasars) at higher redshift than in our vicinity. That's to be expected in the Big Bang theory, since at earlier times, the universe was denser!
You are right, this question is currently under investigation (simply go to www.arxiv.org and search for "SMBH", i. e. super massive black hole). But I don't know why you call this "fudging"...
What "serious flaws" are you talking about? You already listed several assumptions which you consider to be extraordinary (in e-mail), but I don't; but so far, you haven't mentioned any flaws.
Thomas, to what extent do you think the Big Bang is a flawed theory? Do you disagree that the universe was once much, much smaller and hotter than it is now? If so, do you have any explanation for the Hubble effect or the cosmic microwave background?
Incidentally, my understanding was that galactic black holes form by swallowing up gas and dust as the galaxy is forming, and cut off their own growth by a "solar wind" effect where they repel their own food source if they feed too vigorously.
Bjoern
As usual your reasoning is very sensible. I stand corrected, on all of your points especially the one regarding small stars having potentially very long lives. And you are correct, "fudging" is too harsh a word; it implies deliberate tinkering to mislead which I don't accuse the scientic community. But I suspect that current back of the envelop calculations (and there must be some, but maybe not) are not being published because they contradict currently accepted big bang theory. But that isn't "fudging", it is "professional judgment" to investigate further. And you are correct that I view extraordinary assumptions as flaws (e.g. baryiogenesis). As always thank you for your patient and reasoned clarifications.
Morgan
Yes I disagree that the universe was once as small as an atom and thus totally energetic. I know quantum mechanics allows us to think of such high energy denity without matter; but I find it absurd in the same sense that I find general relativitic singularities absurd. There is a point in which every theory excedes its domain of relevance and in my opinion most of the big bang theory resides outside the domain of relevance of either General Relativity or Quantum Mechanics. I don't discount any data; only the various theories through which certain data is (mis)interpreted.
Stephen Hawking once criticized Einstein as follows, "The Einstein universe does not represent the universe we live in because it is not expanding." Well I still think the final details are not in (i.e. too many extraordinary assumptions in big bang theory) and thus the case for a non-expanding universe is still reasonable. I think that we humans are at the very beginning of our understanding of how the universe works and that it is an intellectual lazy to suggest that there are no serious alternatives to the (status quo) big bang theory.
Regarding the CMB in particular. CMB is a phenomenon that had half a dozen credible interpretations in its brief life. I think of the current CMB interpretation (as evidence of the big bang) as just another interim interpretation.
By the way (and this quote by from the 2nd edition of the Encyclopedia of Physics, pg 862 may be outdated), James W. Turan says, "One rather firm conclusion follows from theoretical studies: given the observational limit on the present universal baryon density of the universe and assuming that the microwave radiation is correctly interpreted as a residual 2.7 K radiation temperature, no substantial production of nuclei heavier than He4 is possible in the cosmological big bang." So it seems we've either got a good theory about CMB or a good theory about nucleosynthesis but not both.
I prefer to think of the CMB in the skeptical terms of Victor Weisskopf, "It makes sense to say that an observer is at rest in an absolute sense when the 3 K radiation appears to have the same frequencies in all directions. Nature has provided an absolute frame of reference. The deeper significance of this concept is not yet clear." An old quote but I think still relevant today.
That wasn't actually my question, though. If you're denying that the universe is expanding, I presume you don't think it was ever smaller at all, never mind as small as an atom. So what's with the Hubble effect, the number one piece of evidence for an expanding universe? Or if it's not the idea that the universe used to be smaller and hotter that bothers you, but the consensus on how hot and small it was, what's the cut-off point where your personal credulity can stretch to?
@Thomas:
Your suspection is wrong in two ways: (1) There exist much more than only "back of the envelop calculations" - there exist numerous full-blown simulations; (2) they don't contradict the accepted theory, but are fully compatible with it. As I suggested: look on the arxiv, seach for "SMBH". I posted another comment with several links to papers, but it is currently still held for moderation apparently...
Well, then you must find many experiments done in high-energy colliders absurd, too - since essentially, there we recreate those conditions where one has "high energy density without matter".
And on what do you base that opinion?
If you want to claim that data is misinterpreted, then you have to explain how you arrive at that conclusion. "I think this is not correct", "I think you extrapolate beyond the domain of relevance" or similar statements are not worth anything without evidence supporting them.
If you can explain all the available evidence with a non-expanding (static) universe, go ahead. The available *observational* evidence includes e. g. the following: Hubble law, the abundance of light elements, the existence of the CMBR, the near-perfect blackbody spectrum of the CMBR, the homogenity of the CMBR, the agreement of data on (dark) matter density derived from the CMBR with other data, the agreement of the observed large-scale structure in the universe with simulations of structure growth based on the fluctuations seen in the CMBR; the temperature of CMBR was greater in earlier times; all stars we observed in the Milky Way are at most 13 billion years old; stars in distant galaxies are observed to be younger than the stars in the Milky way, corresponding to the earlier times at which we see them; quasars are observed mainly in a certain redshift range, i. e. they existed mainly at a certain epoch - at an epoch where the universe was denser according to the Big Bang theory, which nicely explains why more quasars existed back then; at great distances, i. e. at earlier times, there were mainly irregular, small galaxies and only very few "mature", big spiral galaxies; the density of dark matter was greater at earlier times; the observed time dilation in supernova brightness curves; Tolman tests.
Additionally, one can prove mathematically both using Newtonian mechanics and General Relativity (Hawking's singularity theorem) that a static universe isn't possible. And again additionally, how do you solve Olbers' paradox in a static universe?
Such an alternative may well exist - but so far, no one has formulated it!
I listed several observations above which *strongly* support the big bang interpretation of the CMBR. You are free to explain all those observations with another theory...
I don't understand what your quote from Turan is supposed to show. No one claims that (many) nuclei heavier than helium were produced by big bang nucleosynthesis! All heavier nuclei were produced later! As Ethan explained in part 7, the observed abundances of primordial elements are perfectly in agreement with the one predicted by big bang nucleosynthesis - so the current theory explain *both* the CMBR *and* the observed elements!
And I also don't understand the Weiskopf quote - only because there is a frame of in which "the 3 K radiation appears to have the same frequencies in all directions", it makes little sense to call that an "absolute" frame of reference. In relativity, when one says that there is no absolute frame of reference, one always means that the laws of nature are the same in all frames of reference, and that one can't distinguish frames of references by measurements done only *within* them. But if you look at the CMBR, you are measuring things *outside* your frame of reference!
Morgan
The question of how much our visible universe has or can contract and expand is a weather problem on a grand scale. It is like asking how much has or can the atmosphere of planet Earth contract or expand. Given the size of planet Earth or the size of the visible Universe, an order of magnitude or few but definitely not 40 or more orders of magnitude as in the big bang model.
One of my big problem with the big bang model of the universe is that it is thermodynamically closed and I suggest that our model of the universe somehow needs to be opened up (e.g. extra dimensions) The particular problems and extraordinary assumptions of the big bang theory (e.g. how much it has or can expand) to me are symptoms of this and other larger problems of the big bang model.
The closed thermodynamic system approximation is a useful approximation to understand systems from atoms to humans to stars to the universe. However the closed thermodynamic system approximation is an assumption that has been stretched beyond its limit of usefulness when it is applied to the entire visible universe.
Everyone agrees that a electron is not a closed sytem; its definition implies an openness in the requirement for an ever present photon field. There is no such entity as an isolated electron (or any other elementary particle). Yet the big bang universe that is somehow built up of such "elementary" open systems is viewed as closed in a fundamental thermodynamic sense (whether it expands forever or collapses in a big crunch or bounces). Something is missing.
In the real world all systems are thermodynamically open system (in the detail); though every system can be usefully viewed as a closed system up to a point. Can the sum of two or 10^80 thermodynamically open "elemementary particle" systems be a fundamentally closed system (i.e. a closed universe)? I don't think so. The big bang model of our visible universe goes many orders of magnitude beyond that point of usefulness of the closed system assumption.
Furthermore, the big bang model of the universe uses the weak cosmological principle that says that from any point in space the universe looks roughly the same. I subscribe to the strong cosmological principle that says from any point in spacetime the universe looks roughly the same (e.g. 13.6 billion years ago, the most distant galaxies observed would still be about 13 billion light years away, and in 10^100 years from now, the most distant galaxies will still be about 13 billion light years away). Neither the weak nor the strong cosmological principle is proven; they are assumptions (hence my bias as well).
Regarding my view of the Hubble effect, it's not appropriate for me to discuss here. pg 30 to 44, follow links to my book online. But I must also acknowledge that Bjoern has pointed out to me several errors in the details of my logic there. He is a physicists, I am a layman.
In the detail, many if not all of my ideas can be refuted; but I think my ideas point in a needed skeptical direction (e.g. regarding the big bang model as the only reasonable theory in town).
Bjoern
All of your points are well taken. I always assume your answers are correct current physics. Thus I suspect that it is frustrating for you that I refuse to accept current big bang theory.
As well I note that the 2006 Nobel Prize in Physics was for the interpretation of the CMB radiation. Thus my skepticism of the CMB interpretation is well outside of current theory.
Further, I acknowledge that in my searches for serious criticism of various aspects of the big bang theory, I find only a few sentences by credible physicists. And rightly so, those few sentences are not enough for you or for me either.
It's really fruitless to argue with the results of high energy or other physics experiments; so I don't. The experimental and observational data is confirmed by others and then must be accepted. But the theory which surrounds the data may be questioned. Though usually high energy physics theory confines itself to narrow interpretations and states its conclusions quite cautiously.
@Thomas:
Yes, obviously. But as I keep pointing out: the big bang theory explains essentially all of the available data (and there is a huge amount of that!), and no other existing theory comes even close to that - by far! There *could* be another theory out there which explain all the data better - but I haven't seen it so far.
I see no big difference in how cautiously particle physicists or cosmologists state their conclusions - neither in their own papers nor in press releases.
First, I don't think this is a sensible analogy; second, I don't see how you arrive at the conclusion that 40 orders of magnitude are "definitely" not possible.
(1) Why is this a problem for you? (2) There are already scores of people working on extra dimensions - what more do you want?
And where is the problem with that? Why can't many open systems together form a closed system?!? And considering that the universe is commonly defined as "all that exists" - how could that be an open system?!?
That is contradicted by *many* observations. I already mentioned several, e. g. the stars in distant galaxies (e. g. galaxies we see at earlier times) are consistently *younger* as the stars in our galaxy; quasars existed mainly in a certain time frame (there are still some quasars today, but most exist at rather high z); the star formation rate was higher in the past; the temperature of the CMBR was higher in the past; the density of dark matter was higher in the past; most galaxies were far smaller and more irregular in the past; etc.
It's a matter of fact that the big bang theory is the *only* theory which can explain all the available data, and I know of no contradicting data. As soon as contradicting data arrives, or as soon as another theory comes along which can explain the available data, I'll admit that the big bang model is not a / the only reasonable theory.
OK Bjoern I respect your opinion but
I don't accept that "the big bang theory is the *only* theory which can explain all the available data". In fact there are an infinite number of theories that can explain a finite amount of data; but you already know that. And Occam's razor blah blah blah.
As for "I know of no contradicting data "; every theory has contradictory data, some minor some major. If there wasn't contradictory data; there would never be a new theory. But you already know that also.
Hence for example, it is out of the contradictory data that perhaps a quantum theory of gravity might be developed; that just might radically change Ethan's "The Greatest Story Ever Told". That is my opinion.
So, I think it is important to remain skeptical especially of the "big bang theory." That is my opinion also, which I am entitled to.
Furthermore, I expect to see many surprises in cosmology and elementary particle experiment, observation and theory in the coming decades. I expect to say many times that I totally misunderstood or misanticipated or misjudged something. But then again, I don't understand things very well.
By the way, I am looking forward to Roger Penrose's upcoming book to be titled "Fashion, Faith and Fantasy in the New Physics, a critique of modern physics". http://www.newscientist.com/article/mg20527511.300-roger-penrose-nonsto…
I hope that he or someone does a better job of critiquing physics than I have otherwise I may have to rewrite my book in 5 or 10 years.
@Thomas:
Yes. But I thought I made it clear that I talked about the *already proposed* theories, not about *possibly existing* theories...?
Yes. That's why I qualified my statement with "I know of...". You quoted that part, but apparently you didn't really notice it...?
Yes. About as skeptical as one has to be of the standard model of particle physics. However, you display *much* more skepticism here.
Well, me too. But in contrast to you, I expect the surprises not in well-established parts of the theories, but in the parts where we still know not much about (i. e. inflation).
Considering Penrose's upcoming book: according to the New Scientist article, he apparently argues mainly against string theory (and I myself am still not sure what to think about it) and against inflation; he prefers a cyclic model (me too!). He doesn't seem to argue against the basic big bang theory anywhere...
ok, dumb question. The galaxies and solar systems do NOT get stretched out with the universe, hence the distance increasing between them. Why then should light wavelengths change ?
and why does the Microwave background radiation remain. shouldnt it have a finite life as the photons pass the observer ? regards
@Mark:
The stars etc. in galaxies are gravitationally bound to each other - light waves aren't bound to each other (or to themselves). So the second do stretch, while the first don't.
The whole universe is filled with the background radiation. Individual photons pass you all the time, but there are always new photons arriving at you, which were emitted at a greater distance and hence take longer to reach you.