"Mirth is like a flash of lightning, that breaks through a gloom of clouds, and glitters for a moment; cheerfulness keeps up a kind of daylight in the mind, and fills it with a steady and perpetual serenity." -Joseph Addison
One of the most brilliant phenomena found in our atmosphere is that of a lightning strike.
Shown below in breathtaking slow-motion, somewhere on the order of 100,000,000,000,000,000,000 electrons are exchanged in a single bolt between the clouds and the Earth's surface.
How does this happen?
Remember that every atom in the Universe -- including in our atmosphere -- is made up of a positively charged nucleus and a whole slew of negatively charged electrons. While we normally think of atoms as neutral, with the number of electrons equal to the number of protons in each atom's nucleus, this is not always the case.
Because, quite often, it's energetically favorable for an atom to become ionized, where it either picks up or loses an electron (or two, or three). Something as simple as table salt is an example of a few ions.
Now, if you can separate these ions from one another, you create a separation of charge, which creates a Voltage. When the Voltage (also known as the electric potential difference) between two regions becomes too great -- even if air is the only thing between them -- it will spontaneously become conductive, and the rapid exchange of charge is what you see as a lightning strike!
You're familiar with lightning happening over great distances, as charge gets transferred from clouds in a thunderstorm down to the Earth itself. But, as I showed you last year, when Iceland's Eyjafjallajökull erupted, oftentimes volcanic eruptions produce lightning as well!
There are some amazing pictures of volcanic lightning that have been shot over the years. Perhaps my favorite is of last year's Eyjafjallajökull eruption, as photographed by helicopter!

While it's been notoriously difficult to catch volcanic lightning in action historically, the feat has been accomplished many times, for many different volcanoes.
For example, here's Chile's Chaitin volcano, from its recent 2008 eruption, its first in 9000 years!
Japan's Sakurajima, an incredibly active volcano in recent history, has been erupting almost continuously since 1955. A Volcano Observatory was set up to continuously monitor its activity in 1960, and has observed volcanic lightning many times, including in this 1988 outburst.
In fact, volcanic lightning has been captured on film as far back as the 1944 eruption on Mount Vesuvius!
Now, I'd love to tell you exactly how volcanic lightning works, but to be completely honest, much as is the case for normal, thunderstorm-based lightning, we aren't 100% sure; it's still an area of active research. (See here and here for some examples.)
But, as a theoretical physicist, I can certainly give you a general picture for what is very likely going on here to cause this, and expand upon what I've previously told you.
Atoms, for the most part, start out neutral. (That's step 1.) But with lots of free energy present, it's certainly no problem to knock the electrons off of some atoms that hold onto them loosely, while at the same time atoms that are keen to pick up these newly freed electrons can do just that. (Step 2.)
That part is absolutely no problem: remember, it's a volcano!
With temperatures of around 1500 Kelvin, there's certainly enough energy floating around to knock electrons off of some of the atoms that hold onto them most loosely, where they can subsequently be picked up easily by other atoms, creating a large number of both positive and negative ions.
Now, the key thing that needs to happen -- from this point -- is we've got to separate the negative charges from the positive ones. (That's step 3.) And we have to separate enough of them, over an interesting enough distance, to get an electric potential difference that will cause a lightning strike! (Step 4.) If we can do that, we can make volcanic lightning.
So how could we separate these charges? Remember what we've got here: a bunch of ionized atoms -- both positive and negative ions -- in a hot, turbulent environment. Coming up from the depths of the Earth, we should have a great many elements of interest involved here.
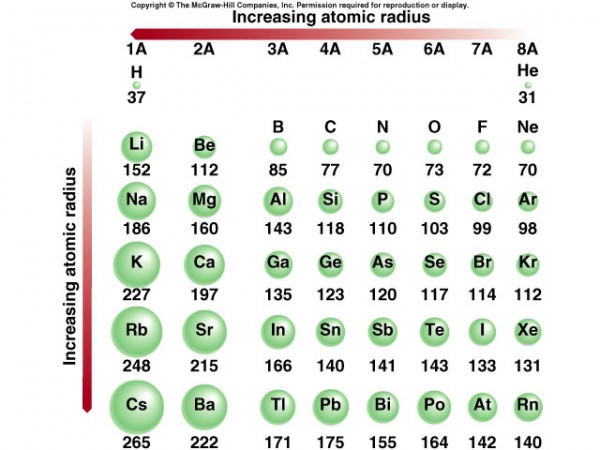
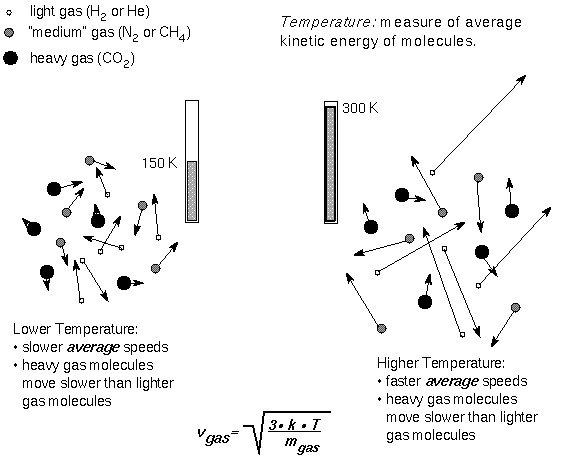
Right away, one of the first things you notice about these elements is that they have different masses from one another, as well as different radii! Now, they should all come out at a high temperature, which cools over time, once they leave the volcano itself. This is very important for the speeds of the atoms/ions in question.
In general, when the atoms and ions first come out, they're moving more quickly, and they cool over time, slowing down.
That's no big deal on its own, but there are two other very important factors that make it very easy to separate these positive and negative charges from one another. First off, these ions have very different masses from one another!
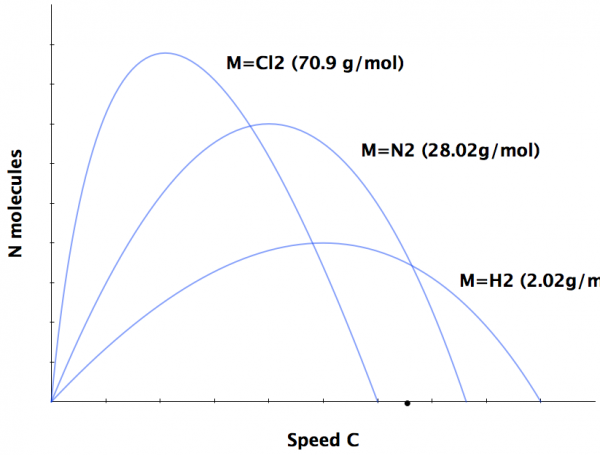
The heavier the atomic weight of an element is, the slower it moves, even at the same temperature as a lighter element! That means a lot of different things, including that heavier ions have more inertia, and it's more difficult to change their momenta. So these slow-moving, heavy ions will move around very differently to fast-moving, light ones. Moreover, this is true at all temperatures!
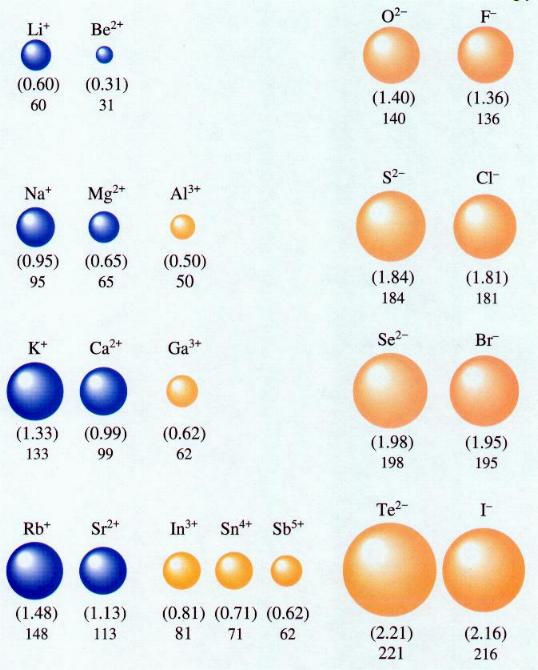
The second very important factor that makes it easy to separate these ion-types from one another? The tremendous difference in sizes -- and hence cross-sections -- between positive and negative ions.
Sure, elements have different sizes from one another in the fashion I showed you above. But ions work in a much more dramatic fashion! Let's take a look at exactly how.
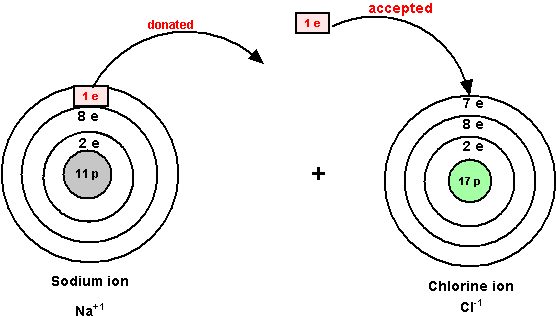
In general, negative ions are huge, and positive ions are tiny! Why is that? You put more electrons on your atom, and they repel one another; the nucleus (with fewer protons than there are electrons in this ion) cannot hold the electrons as tightly as it could if the atom were neutral, and the atom increases in size. On the other hand, to become a positive ion, you kick electrons out of your atom, and the nucleus (with more protons that there are electrons in the ion) holds onto the electrons more tightly than before!
This means that the negative ions have larger cross-sections than the positive ions, and hence they interact very differently than the positive ions do!
Combine these things together: different mass ions moving at different average speeds with different cross sections in an environment with a temperature gradient, and there's your charge separation! And what does that give you?
Volcanic lightning! The above, and all the subsequent photos, are of this June's Chile ash cloud, seen both over Chile and Argentina. These are some of the most recent -- and most spectacular -- photos of volcanic lightning ever taken. Enjoy!
And as an added bonus, those of you on google+ can check out an entire album of volcano/volcanic lightning photos here!
















Impressive!
I thought we were talking about ash particles, not ions. Are there actually Mg++ ions floating around by themselves? This seems unlikely to me, that kind of charge density is going to attach to something.
The Bobs,
Of course, one of the dangers when you are a theorist is that your "simple" model may in fact be oversimplified, and that may be the case here, for reasons you have intuited.
In reality, the size of ash particles varies tremendously, from sub-micron sizes up to many millimeters, and typically carry much more charge than just a single electron or two. (See here for a discussion of ash-particle size.)
But the overall argument -- different masses with different charges at different mean speeds, in the presence of a temperature gradient -- still applies, although I have no doubts that the quantitative details are vastly different (and far more complex) than the simplified analysis I've presented here.
Still, thanks for calling me on it!
These are some of the most spectacular images I have ever seen. Thanks for posting them. I live on an active volcano and have seen a few small displays, but nothing as spectacular as these.
However, at least twice, something really strange came out of my volcano (Volcan Tungurahua, Banos, Ecuador): BALL LIGHTNING!!! Once I saw it myself, and another time I didn't see it but it was filmed by the national news. Of course, the news program concluded that it was a UFO, but I am sure it was ball lightning. It is a glowing sphere that very slowly rose out of the crater, hovered above it (more or less stationary) for maybe 30 seconds, and then slowly drifted away horizontally.
Back when I was in physics, ball lightning was quite mysterious, and some people even doubted that it exists. Do we know anything more about it nowadays?
I love these pics. When I first saw pictures of the Chilean eruption a few years ago I was sure they were fake at first. If I'd seen these scenes in a movie I'd call it unrealistic and over the top.
The real world is so much cooler than all the pseudo-science and fiction people feel compelled to create for themselves.
One thing, about those Eyjafjallajökull photos from Marco Fulle. I was wondering right away how they would manage to stabilize a camera on a helicopter for any length of time more than 1/20 s or so. The, I noticed star trails in the background! And I started having my doubts. So I checked the link, and it says "terrestrial photos" as opposed to other folders labeled "aerial photos", so I think you should probably correct the bit about "as photographed by helicopter."
Randy,
I'm pretty sure the photos by Marco Fulle are, in fact, by helicopter. First off, I don't think there are any sites in Iceland nearby Eyjafjallajökull that are at the necessary elevation to capture a shot like the one he has.
Second, all the secondary sources I can find have very detailed reports of Marco Fulle taking these photos via helicopter (see, e.g., this report, as well as this one).
They are spectacular, no doubt, and I don't know how he pulled them off technically, but I would be shocked to get confirmation that Marco's photos were not, in fact, taken via helicopter. (Of course, if you can provide that confirmation, I'll quickly change it to reflect the reality!)
Sometimes, when you see spectacular images of fast-moving events shot by professional photographers, what you're really looking at is a carefully chosen frame from a movie. (The distinction between "still" and "movie" has been steadily blurring for a long time) Some popular nature photographers use 70mm movie cameras in order to take advantage of film latitude for cinema film and the various lenses available. If you point a 70mm movie camera at an otter doing just about anything, and shoot 30 seconds of film, you're guaranteed at least one printable frame of adorable cuteness. Decent digital SLRs nowadays have movie modes (like my Canon 5d) that basically give you a stream of images through the lens at full resolution; they're comparable to 35mm frames shot at 30 frames/second. I'm not sure if that's how some of these shots were done, but if I were trying to capture lightning, explosions, soccer goals, etc, that'd be how I'd do it.
This is a video by another photographer showing volcanic lightning from last year's eruption in Iceland.
@Marcus: That's great for daytime shooting, yes. But all those awesome pictures you see of lightning (volcanic or otherwise) are almost always made by leaving the shutter open for a longer time, at night. You don't get that much lightning going off all at once; by leaving the shutter open for a while, you get the combined effect of several lightning strikes on one image.
There is the alternative of taking a movie and stacking the frames. But then, either you're picking out the good frames (and the star trails wouldn't be so continuous) or you're stacking them all, which doesn't really differ much from taking one long exposure.
I'm going to agree with Randy here and guess that Marco's images in this post were shot from the ground, zoomed in as much as his equipment would allow. The news reports do claim that Marco shot pictures from a helicopter, but considering the source (the NY Post is not entirely reliable, and it is not independent of Fox News as both are owned by Rupert Murdoch), my guess is that the journalist garbled the information: some of Marco's pictures were indeed shot from a helicopter at the claimed distance from the volcano, but not these photos. Other photos he shot of this event were clearly taken from the ground, including one lightning photo where you can see the highway that runs through the area--I can't be sure, but it looks like it may have been the same direction as the photos Ethan shows.
"We did it, Mister Frodo--we made it to Mordor."
These would be great desktop wallpapers.
Amazing stuff. Still, I'm confident we'll clear up this chaos-theory detail fairly soon.
A more interesting problem, at least in my view, is the following: Can photons attain any frequency?
My question isn't really about lower and upper bounds, but let's start there.
Upper bound: Since the energy of a photon is E = hv and since the Universe presumably doesn't have infinite energy, there clearly must be an upper bound on a photon's frequency. Do we know what this bound is?
Lower bound: I have no information to guide me here except to say that a photon with a frequency of 0 probably can't be considered a photon. Do we have any better lower bound?
And now to my real question. A photon can be created by an electron in an atom falling from a high energy state to a low energy state. But these states and their energy levels are discrete and therefore no matter how many types of atoms or molecules you have, the photons thus released can only cover a finite, discrete range of frequencies.
OK, but there are other ways to make photons. Such as nuclear reactions. I'm not too famaliar with these reactions, so can you help me here as to whether they can produce a photon of any desired frequency?
Lastly we have the expansion of the Universe. Photons sent off when the Universe was young have had their frequency reduced due to spacetime expansion. Does this mean these photon's frequency, as recieved here on Earth, now cover all infinite types of frequencies? I personally don't see how.
Here's hoping you will answer my question! :-)
The Chilean volcano is called Chaitén, not Chaitin.
Thanks for credit my image. Appreciated
thanks for the images