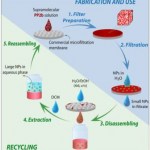
nanoparticle filter
Two completely unrelated papers have got us thinking about chemical bonds. When we refer to chemical bonds, we generally mean covalent bonds: Atoms become "wedded," sharing electrons, and breaking them apart takes energy. By comparison, other types of bonds are weak attractions - mere flirtations, or mild sparks between hydrogen and oxygen in passing water molecules.
So why would a researcher in organic chemistry - a field based on carbon, the king of covalent bonds - be investigating the properties of non-covalent, hydrophobic bonds? The answer, of course, is that they can be used to create…