tags: researchblogging.org, Brown paper wasp, Polistes fuscatus, hymenoptera, evolution, eusociality, social behavior
Brown paper wasp , Polistes fuscatus.
Fairport, New York, USA. 2003.
Many thanks to Alex Wild for sharing his amazing images here.
Thanks to Elizabeth Tibbetts for the species identification.
[larger view]
Eusociality, or "true social behavior", is the most extreme form of cooperative sociality known. Due to its seemingly altruistic nature, eusociality has provided many interesting challenges for evolutionary theory. Eusociality, as exemplified by ants, bees and wasps, is defined as a cooperative living situation where a significant portion of a genetically related group, often known as "workers", spend their lives caring for their siblings instead of reproducing.
But how did eusociality come about in insects? It has been suggested that eusociality has a genetic basis that was evolutionarily modified from already extant maternal behavior. However, this gene-behavior connection has not been definitively established, so eusociality could instead have evolved from reproductive behavior. Further, many questions remain as to the precise mechanism for how eusociality evolved.
To investigate the molecular mechanism underlying the origination of eusociality, a group of American scientists led by Amy L. Toth, an entomologist at the University of Illinois at Urbana-Champaign, compared brain gene expression patterns found in different groups of the primitively eusocial hymenopteran, Polistes metricus, which is a close relative to the wasp pictured at the top.
Polistes is considered to be primitively eusocial because both workers and reproductive individuals show maternal (foraging and provisioning) and reproductive behaviors at different times during the lifetime of a colony. For example, foundresses are female Polistes wasps that establish new colonies in the spring and, because they are often alone at this time, they must necessarily show both reproductive and maternal behaviors. However, after the first generation of offspring reach maturity, the foundress then becomes a queen, whose job is to focus exclusively on egg laying, while she relies on her original offspring, who are workers, to care for their siblings by foraging and feeding them. Late in the season, gynes appear in the nest, but don't contribute to either maintenance or brood care. Instead, their job is to mate, successfully overwinter and then establish a new nest the following spring. As you can see, primitively eusocial insects do show some overlap in their division of labor, and additionally, they lack physical differences that distinguish the different groups.
In contrast, advanced eusocial insects such as honeybees, have different castes with distinct morphologies and specific associated behaviors -- either foraging/maternal behavior or reproductive behaviors -- and these castes virtually never overlap. But Polistes is a wonderful research organism for gaining understanding into the molecular origin of these behaviors because their behavior changes over time, and thus, it is proposed that their brain gene expression patterns likewise will change.
Because the researchers were interested to learn about the genetics underlying social behaviors, they decided to determine whether maternal or reproductive behaviors have a greater shared molecular basis with social behavior. Further, because most behaviors, even those shown by seemingly simple insects, are complex and polygenic traits, it was necessary to analyze a large variety of genes that participate in numerous molecular pathways.
To make comparisons between the genes underlying these behaviors in Polistes, the team compared changes in brain gene expression patterns that characterized the four distinct groups. Based on behavioral information in these insects, the team proposed two hypotheses; either maternal or reproductive behavior would be most similar at the molecular level regardless of which group was showing the behavior, and would therefore show more commonalities in gene expression patterns. Thus, if maternal behavior was most similar in foundresses and workers, then the gene expression patterns seen for each of these groups would closely resemble each other. On the other hand, if reproductive behavior was most closely related between the queens and foundresses, then these two groups' gene expression patterns would be most similar. Gynes, of course, being the most behaviorally unique should also have the most diverse gene expression patterns in their brains.
To do this work, the team measured brain gene expression in 87 individuals from
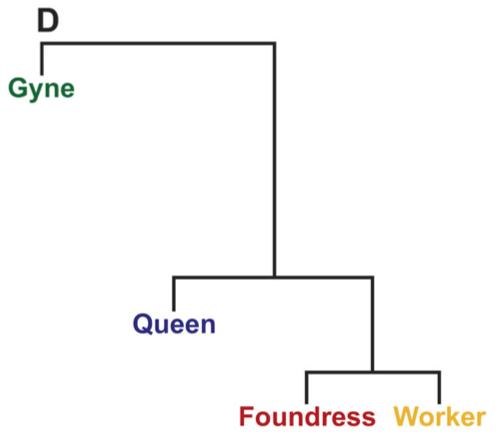
four distinct behavioral groups of females from naturally occurring colonies of Polistes. After conducting a variety of analyses (the most straightforward of them is pictured below), the team found that brain gene expression patterns of workers and foundresses was most similar, as demonstrated by the shorter branch lengths between these two groups. Interestingly, foundresses and queens, who both show reproductive behavior, has fewer similarities in gene expression patterns with each other than foundresses had with workers. Predictably, gynes were most distinct group of all, as revealed by the much longer branch lengths in this phylogeny (Fig. 1D, below),
These data reveal that, not surprisingly, workers had more similarities in brain gene expression patterns with foundresses, who also show maternal behavior, than they had with either queens or gynes, which do not. Thus, this helps establish a connection between specific behaviors and the expression of a particular group of genes. Further, these data suggest that as foundresses become queens, they undergo a dramatic alteration in their brain gene expression patterns, which then affects their behavior profoundly (of course, one can also make the same claim for the changes experienced by the gynes as they become foundresses).
So in short, this research demonstrates a definite connection between gene expression and behavior: that particular behaviors are associated with specific patterns of gene expression.
Sources
"Wasp Gene Expression Supports an Evolutionary Link Between Maternal Behavior and Eusociality" by Amy L. Toth, Kranthi Varala, Thomas C. Newman, Fernando E. Miguez, Stephen K. Hutchison, David A. Willoughby, Jan Fredrik Simons, Michael Egholm, James H. Hunt, Matthew E. Hudson, Gene E. Robinson. Science DOI: 10.1126/science.1146647 (September 27, 2007) [abstract]