The laws of thermodynamics are empirical laws - they were not derived from some first principles of the universe: they were derived by doing thousands and thousands of experiments, and then coming up with some relationships that could quantitatively explain all those experiments.
In biological thermodynamics, we are at the beginnings of trying to define a similar set of "laws of biothermodynamics" - in this case we want relationships that connect thermodynamic quantities (ÎG, ÎH, ÎS, ÎCp) to functional or structural information about the biomolecules themselves. Nobody has anything that…
biophysical
Scienceblogs is promoting the writing of "Science 101" general topic posts all through the "back to school" month of September. So, here is the first in a multi-part series on Heat Capacity in Biology:
Heat Capacity in Biology 101: What is it?
Heat capacity is basically a proportionality constant. For any substance, the heat capacity tells you how much the temperature of the substance will change when you add a specific amount of heat.
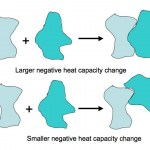
Here is an absolutely beautiful schematic illustration of the difference between a small heat capacity and a large heat capacity (from a website on the…