"I like cappuccino, actually. But even a bad cup of coffee is better than no coffee at all." -David Lynch
Particularly in the dead of winter, most of us enjoy a hot drink, whether it's coffee, tea, hot chocolate or soup. But if that drink is too hot, your options for cooling it down are unsatisfying: wait for the room to cool it, which takes forever, drop an ice cube in, which waters down your drink, or blow on it.
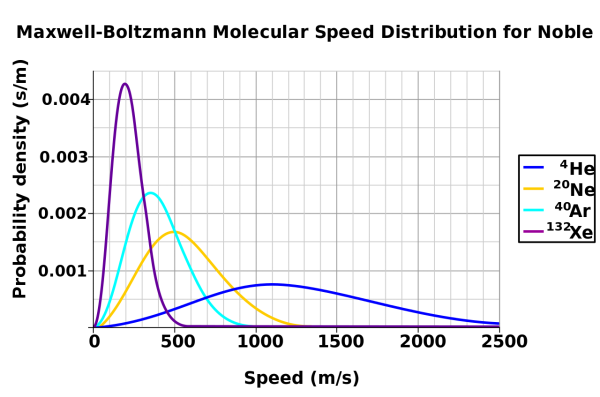 Image credit: Wikimedia Commons user Pdbailey, who created this image and placed it in the public domain. The speed distribution is qualitatively the same for liquids as it is for gases.
Image credit: Wikimedia Commons user Pdbailey, who created this image and placed it in the public domain. The speed distribution is qualitatively the same for liquids as it is for gases.
While blowing on your drink may seem ineffective, as the breath inside your body is generally warmer than the ambient air, there's an additional feature that makes blowing on it totally worth it: the circulation and exposure-to-air of the hot fluid vastly increases the rate of evaporation, taking the highest-kinetic-energy molecules out of the equation and cooling your drink more quickly.
Go read the physics behind why blowing on your hot drink actually does cool it down faster!


The same process, evaporative cooling, is important in atomic, molecular and optical physics (AMO) for production of extremely low temperature systems, such as Bose-Einstein condensates.
In a typical application, a collection of relatively slow (low temperature) atoms are contained in a trap of electric and magnetic fields. The system is tuned in such a way that the fastest moving atoms can escape out of the trap (and stick to the walls), while the slow ones are turned around and kept in place by the field configuration.
Over time, you end up with a fewer atoms, but the collection is all together much colder (nanokelvins or below).
I'm not an expert on this, but I think the rate of evaporative flux from a liquid to surrounding fluid is independent of the properties of that surrounding fluid. But there is a return flux (into the water) from the fluid, and this is increased if the partial pressure of water vapor is increased. So by blowing on the surface, you've reduced the backflux of water vapor into the water, thus reducing net evaporation.
One shouldn't really talk about a given molecule being hot/cold, temperature is a statistical property, and a single molecule taken in isolation doesn't have enough thermal degrees of freedom to determine it.
Actually, your hot drink is a micro version of the universe's entropy on display at micro level.
https://en.wikipedia.org/wiki/Heat_death_of_the_universe
Blowing the hot beverage in reality doesn't cool it that much. It only slightly cools the topmost layer in the cup.. or spoon.. the top 1-2mm. The reason being that air is a very poor heat conductor. If blowing was the only means to cool the cup of coffee.. we would be blowing it for hours and hours.
The dissipation of heat through the walls of the cup is much more significant in terms of overall cooling of the drink then blowing the top is. Simply because the surface area is larger.
A good blow across the cup's surface (rather than directed down into the liquid) is also going to create a Bernoulli effect; i.e. your rapidly moving breath (slightly) lowers the pressure above the surface, leading to (slightly) more evaporation.