It's been a while since I've done a post over-analyzing some everyday situation, because I've been too busy to do any silly experiments. We're on break this week, though, so I took a little time Monday to bring excessive technology to bear on the critically important scientific question: how good is my insulated Starbucks cup?
To back up a little bit, because it's always important to provide background and motivation when writing a lab report, I spend a lot of time working at Starbucks these days, because when I try to get work done in my office, people keep showing up wanting me to do stuff for them. Being a good squishy liberal type, I started to feel guilty about the number of paper cups I was using to drink my tea (it's a little ironic that I spend all this time in Starbucks, since I don't like coffee), so I sprang for a fancy insulated cup, seen at the left in the "featured image" above (RSS readers, you'll need to click through). This turns out to be remarkably good at keeping my hot caffeine hot, way better than the old insulated mug I got as a conference freebie ten years ago, so I've been toying with the idea of doing an experiment to figure out how good it is.
Since I'm an academic physicist, I just raided the teaching labs for two Pasco temperature probes, and hooked them into DataStudio to record the temperature as a function of time. This ended up producing a data file that was kind of silly-- the lowest sample rate I could set was 1Hz, which is ludicrous for a slow thermal process-- but on the plus side, it didn't require me to actively monitor the experiment. As a control of sorts, I used the Insititue for Quantum Computing water bottle I got during my visit to Waterloo last fall. These are almost the same size, and both are made of metal. The chief difference between them is that the Starbucks cup is built like a thermos bottle, with at least two layers of metal separated by an air gap. The IQC bottle is a single thin metal wall.
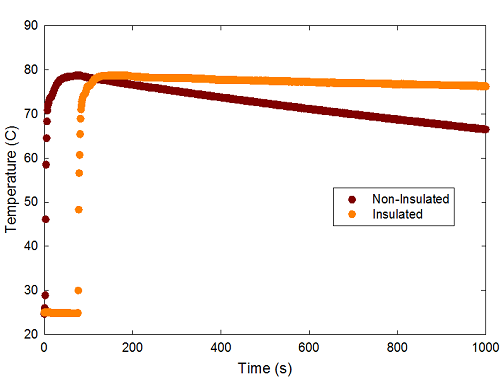
The initial stages of the experiment can be seen here:
This shows the temperature as a function of time for the two containers. I filled the IQC water bottle first (using the Starbucks cup to transfer hot water from the water cooler in the department office; the IQC bottle is slightly bigger, and this guaranteed they would each contain close to the same amount of liquid), then went back and filled the Starbucks cup. As a bonus, this gives a nice baseline value for room temperature (about 25 degrees C, as you can tell from the initial points for the insulated cup, befor eI put the probe in the hot water). You can see that there's an initial rapid increase, followed by a slow cooling down.
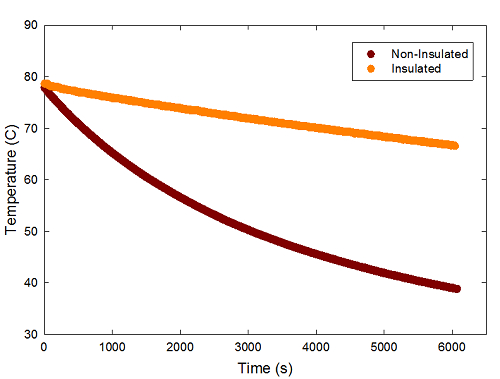
The graph above cuts off after a fairly arbitrary 1000 seconds. The real test of the two, though, demands a much longer run, so here's a cleaned-up version showing just the cooling period:
Here, I've cut off the initial increase, and started the two at the first point where they reached the maximum temperature recorded for each (77.9 C for the IQC bottle, and 78.7 for the Starbucks cup). I then let this run for a bit more than 6000s, tracking both temperatures. Why 6000s? Because at that point, it was time for me to go to the gym, and then home to do other stuff, so I stopped the experiment at that point.
As you can see, the insulated cup clearly cools off much more slowly than the non-insulated bottle-- after more than an hour and a half the water in the IQC bottle is down to about body temperature at the end of this, while the water in the Starbucks cup was still over 65C, distinctly hot. So, it's quite clear that the insulation makes a big difference.
Can we be more quantitative about this? Of course we can-- that's the whole point of making the pretty graphs in SigmaPlot, after all... The temperature decrease we see here looks very much like an exponential decay, so I fit it to one. Left to its own devices, SigmaPlot prefers these to asymptotically approach temperatures well above room temperature-- about 30 and 45 degrees for the IQC and Starbucks cups, respectively. But since I know the temperature of the room from those baseline points, I just forced it to approach a final value of 25 C, and got the characteristic decay times for the two containers.
The non-insulated IQC bottle cooled with a characteristic decay time of 4,411+/-4s, or about 1.23 hours. The insulated Starbucks cup cooled with a characteristic decay time of 24,560+/-12s, or about 6.82 hours. That's the time taken for the difference between the temperature of the cup and room temperature to decrease by about a third, or down to around 41 C. In other words, if I got a full cup of tea first thing in the morning, it would still be at least lukewarm at the end of a working day.
Now, there are some obvious caveats to this. Despite the really small uncertainty values I quote above, I wouldn't put too much stock in those fit values, because it's really fitting only a tiny part of the decay. You'd really like to see it go for at least one characteristic decay time, in order to have full confidence in the fit results-- as I said above, unless I forced the fit to approach the known value of room temperature, it wanted to assume a much higher asymptote. The error was smaller for the IQC bottle, which went through almost one and a half decay times than for the Starbucks cup, which only got through about a quarter of one. This isn't a complete show-stopper, of course-- we know the decay rates of radioisotopes with million-year half-lives, after all-- but it would require a bit more care than I took here to really do it right. There are also some slightly dodgy assumptions baked into this regarding the mathematical description of the cooling, and the constancy of room temperature. But on the whole, this showed what I expected: the Starbucks cup really is very good at keeping my hot drinks hot.
The way to improve this measurement is obvious: wait a longer time. There's a small technological issue, though, in that I don't know if DataStudio is stable enough to record 30,000 data points without crashing. Of course, there's no need to sample it every second-- every five or ten minutes would be plenty for these purposes-- but that would require me to use either a more or less sophisticated system to record the temperatures, and I just don't care that much.
So, anyway, there you go: another post over-analyzing everyday physics. Because that's how we roll in the experimental physics community.

It looks like you were able to keep the lid on the thermos and just fit the probe through the drinking port, whereas the water bottle had to be propped open slightly. I doubt this matters much for the bottle, since the heat transfer through the metal would probably be more efficient than through the air above the liquid, though loss of vapor could have a pretty significant effect on the rate of cooling.
When you repeat the experiment, you should do multiple trials with the lids closed, and measure the final temperature at different time points (since a wireless probe is probably too fancy).
/science pedantry
How does probe insertion impact the insulating and/or sealing properties of the lid? Looking at my Thermos travel mug, I don't see a good way to practically insert a probe without removing the lid entirely, which would likely invalidate the procedure. I don't know how different the Starbucks mug might be.
The hole in the lid of the Starbucks cup is, indeed, large enough to stick the Pasco temperature probe through. It's a little tight, but not too bad, and only lets a small amount escape. The IQC water bottle had the lid off, because there's no hole in it.
I don't think the gap at the top is all that big an effect, but it's a little hard to say. That could be nailed down by sealing it up between measurements at large intervals, but again, I don't want to have to babysit an experiment I'm doing just for the blog.
This is a great experiment, and something that is begging to be done as a science fair project.
You can get USB dongle dataloggers for $50-60 now (Omega sells a five pack for $160!), which can be droppedinto the containers and left for as long as you want (no babysitting required).
Multiple runs, with either "bare room temperature" or controlled environment (e.g., a refrigerator) would let the student see whether the cooling rate depends on the external baseline.
Besides insulated or not, how does a proper dewar flask (Thermos) perform? Does extra insulation (like a beer cozy) improve the plain metal bottle, or the insulated cup?
This measures the undisturbed cup pretty well, but I don't drink from an undisturbed cup. (And while I do like coffee, I drink tea during the day.) You need an apparatus that pours out a small volume (20 ml?) every few hundred seconds. The interesting difference is the last deciliter in the bottom of the cup!
Michael, it just so happens that my son's science fair project this year is looking at the heat-retaining properties of various hot beverage cups, both disposable and otherwise. He's collected the data already but hasn't put it all together, but the most interesting preliminary finding is that my travel mug allows the beverage to cool quickly to the high end of "optimum hot beverage range", and then it very slowly cools to the bottom of the range before rapidly cooling to room temperature. This takes the famous Thermos question ("It keeps hot hot and cold cold: how does it know?") to a whole new level.
@Christopher Bohn -- even more awesome :-) What did your son use for DAQ? Also, echoing "tc>"'s comment, was the cooling measured in a full container left undisturbed? Or was typical drinking modelled?
The varying cooling rates in the travel mug is interesting!
Upon seeing the beauty of the exponential curves generated by the data, I weep sir, I weep.
My son's data acquisition was done with a kitchen thermometer. The cups/mugs were left full -- I suppose you could say that the drinking model was "set it down and forget about it until after checking emails and reading the news".
I just looked at his chart, and my travel mug's drop-off below 120F isn't as pronounced as I remember it being at the time, but it definitely stayed in the optimal range longer than any of the other cups/mugs tested.
Re: Christopher Bahn - I wonder whether that travel mug is equipped with beeswax or the like, which has a phase change at about the right temperature for hot coffee. That's apparently how Coffee Joulies work.
I see a slight inconsistency here in the handling of the bottles: "I filled the IQC water bottle first (using the Starbucks cup to transfer hot water from the water cooler in the department office; the IQC bottle is slightly bigger, and this guaranteed they would each contain close to the same amount of liquid), then went back and filled the Starbucks cup." So when the IQC bottle was charged with hot water, the bottle was at room temperature. When the Starbucks cup was filled for the test, its inside had already absorbed some heat from the water transferred to the IQC bottle. This should make no difference to the curves, but the bottles will have slightly different starting temp points. Assuring equivalent initial conditions would be important for a really accurate result but obviously it's not in this case.
I work with industrial lasers, often in places where you can't readily get a good cup of tea. A colleague and I discussed fabricating an adapter between our company-issued thermal mugs and the laser power delivery fiber so we can bring water to a boil for our favorite beverage. Joking about having a $250,000 Yb-YAG 'kettle' got that idea scotched in a hurry.
I was very interested in reading this post, and I was happy to see how hot my Starbucks cup will keep my drink. Other comments have said things about the lid being open affecting the results, but if you’re drinking your drink wouldn’t the lid need to be open? Maybe I’m not exactly concerned with accurate numbers, but I’m going to be drinking my drink, and not just letting it sit until it gets cold.