tags: virology, mimivirus, sputnik, virophage, microbiology, molecular biology
Now here's an astonishing discovery that's hot off the presses: a virus that infects other viruses! This amazing finding is being published tomorrow in the top-tier peer-reviewed journal, Nature.
I don't know about you, but when I was in school, I was taught that viruses could only infect other living cells, and further, I was taught that viruses are not living cells. So, logically, one could conclude that viruses cannot infect other viruses. But a new discovery by a group of scientists in France reveals otherwise.
The story began in 2003, when Didier Raoult and his colleagues at the Universitee de la Mediterranee in Marseilles, France, discovered Mimivirus in a water cooling tower in Bradford, UK. That virus is so huge -- its linear chromosome is so large that it has the capacity to encode more genes than many bacteria and archaea species, for example -- that it challenges the accepted definition of what is a virus. Mimivirus primarily infects the amoeba, Acanthamoeba polyphaga, although antibodies to this virus have been detected in some humans recovering from pneumonia, suggesting that it might have infected humans as well. Mimivirus is known as Acanthamoeba polyphaga mimivirus, or just APVM.
Recently, Raoult and his colleagues were visiting another water cooling tower, this one in Paris, when they found a new strain of mimivirus there. To their surprise, while examining their new find with electron microscopy, it was even larger than the original mimivirus, so they christened it mamavirus. But their surprises did not stop there because they also saw a lot of tiny icosahedral viral particles, a mere 50nm in size, attached to the giant mamavirus (figure 1);
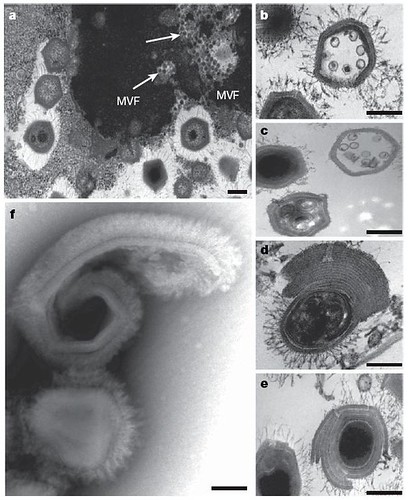
Figure 1 | Different morphological aspects of mamavirus and Sputnik.
a-e, Observations by transmission electron microscopy; f, observation by negative staining electron microscopy. a, Mamavirus virus factory (MVF) with mamavirus particles at different stages of maturation. Clumps of Sputnik particles (arrows) are observed within MVF. b, In some cases, Sputnik is observed within mamavirus capsids. c, Defective particles are produced. d-f, Co-infection with mamavirus and Sputnik results in abnormal morphology of mamavirus particles, such as membrane accumulation at one side (d), membrane accumulation around the particles (e), or open particles (f). Scale bars, 200 nm [larger view].
Thinking this was another virus that infects amoeba, the researchers injected the tiny virions into amoeba. But the tiny virus did not multiply. But amazingly, this virus, which the researchers had named "Sputnik" due to its shape, did multiply when injected amoeba along with either mimivirus or mamavirus (figure 2);
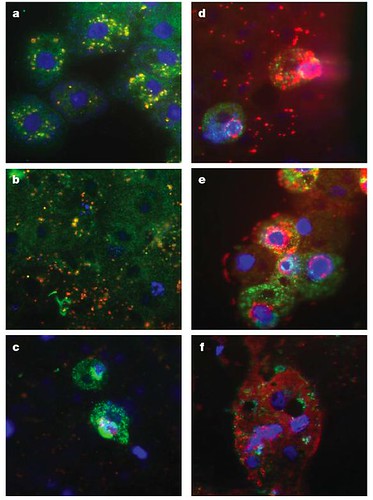
Figure 2 | Sputnik propagation in mamavirus-infected amoebae.
A. castellanii cells were infected with a mixture of mamavirus and Sputnik. Indirect immunofluorescence labelling was performed with rabbit antimimivirus serum (red) and mouse anti-Sputnik serum (green), and nucleic acids were stained with 4,6-diamidino-2-phenylindole (DAPI; blue). a, Numerous Sputnik virions entered the cytoplasm at 30 min after infection. b, At 4 h after infection, the first viral factories were seen as distinct, strongly stained patches. No viral particles could be seen in these cells, indicating an eclipse phase. c, At 6 h after infection, the viral factories expanded and were homogenously and strongly stained with DAPI. Sputnik production was detected at one side of the viral factory, but no mamavirus virions. d-f, At 8 h after infection (d), mamavirus production was observed; this increased extensively at 12 h (e) and 16 h (f) after infection [larger view].
They found that Sputnik co-infection resulted in the production of damaged the mamavirus virions so they were not infective, and also dramatically reduced the probability that infected amoebas would burst open -- unlike the normal progression for a mamivirus infection. Thus, it was concluded that Sputnik actually infects mimivirus or mamavirus. Because Sputnik's behavior is so similar to the effects of bacterial viruses, or bacteriophage, upon bacteria, the researchers refer to this new type of virus as a "virophage", and suspect it may represent a new virus family.
"Sputnik reproduction seems to impair the production of normal APMV virions significantly, indicating that it is a genuine parasite," write the scientists. "We have therefore termed this virus a virophage by analogy with bacteriophages; should other similar agents be discovered in the future, virophage could be used as a generic name to denote them."
Sequence analysis reveals that Sputnik's circular double-stranded DNA genome consists of only 18,343-base-pair (bp) with 21 predicted protein-coding genes that range in size from 88 to 779 amino-acid residues (figure 3). Compare that to its probable host, APVM (mimivirus), which has more than 900 protein-coding genes.
Figure 3 | The Sputnik chromosome.
The predicted protein coding sequences are indicated on the two DNA strands (first, outer, circle) and coloured according to their corresponding homologues. ORFs with homologues to mamavirus/mimivirus are indicated in blue, ORFs with homologues to other NCLDVs and bacteriophages are shown in green, and the ORF homologous to an archaeal virus gene is shown in red. The virion protein coding sequences are shown in purple and ORFans are shown in grey. Phylogenetic trees are displayed for the predicted protein coding sequences with homologues in nr and/or the GOS data sets along with the 2D-gel identifying the capsid protein. GC skew and G+C content are indicated in the second and third circles, respectively. IPG, immobile pH-gradient buffer [larger view].
Sputnik is also interesting because 13 of its 21 encoded proteins are "ORFans." ORFs are "Open Reading Frames" meaning that those regions of the genome probably encode a gene product of some sort, so ORFans are genes that have no detectable homologues in the current sequence databases, meaning that we cannot even guess their function based on their similarity to other genes that we've already identified and studied.
However, phylogenetic analyses of the remaining eight genes indicate that they have viral/plasmid, bacterial or eukaryotic homologues, or homologues from the environmental Global Ocean Survey (GOS) data. GOS sequence data were retrieved from the world's oceans by Craig Venter in 2004 and are thought to be comprised mainly of microbial DNA sequences. Thus, the Sputnik genome contains eight genes that are evolutionarily related to at least three distinct sources: a proposed novel family of viruses; either archaeal viruses or plasmids; and third, either mimivirus or mamavirus.
Could Sputnik infect humans, or alternatively, might scientists discover similar virophages that we could use as therapeutics because they infect those viruses that infect humans? The researchers speculate that size plays a role in whether a virus can be infected by a smaller virus such as Sputnik. Unfortunatly, medium-sized viruses such as HIV and avian influenza are too small to be infected, while the extremely large mimivirus and mamavirus can be. It makes me wonder if the extinct large virus, smallpox, might have had its own virophage(s)?
Source
Bernard La Scola, Christelle Desnues, Isabelle Pagnier, Catherine Robert, Lina Barrassi, Ghislain Fournous, Michele Merchat, Marie Suzan-Monti, Patrick Forterre, Eugene Koonin & Didier Raoult. The virophage as a unique parasite of the giant mimivirus Nature DOI:10.1038/nature07218. .


OK, this shit is gonna take some more thinkin'.
I can't get the paper at home, but i'll be all over it in the morning.
[Carson] That is just wild! Did you know that, Ed? [/Carson]
[McMahon] YES! Ha, ha ha! [/McMahon]
So the virophage is hijacking the ameba's innards to reproduce itself first, and secondly to take a rival virus out of the running without killing the host?
Hey, in this day and age, you make a living any way you can.
So the virophage is hijacking the ameba's innards to reproduce itself first, and secondly to take a rival virus out of the running without killing the host?
Um, no.
"Thinking this was another virus that infects amoeba, the researchers injected the tiny virions into amoeba. But the tiny virus did not multiply. But amazingly, this virus, which the researchers had named "Sputnik" due to its shape, did multiply when injected amoeba along with either mimivirus or mamavirus."
It only infects the m[a,i]m[a,i]virus, not the amoeba.
sputnik is a virus, so is only able to replicate itself when its host, mamavirus (mimivirus) replicates itself. but mamavirus (mimivirus) is also a virus, so it can only replicate itself when infecting a living cell, an amoeba. so as a result, the only way that sputnik can replicate itself is when its host, mamavirus (mimivirus), is replicating inside an amoeba .. .. does this make sense?
Fascinating! Especially fascinating to me is the statement that humans had developed antibodies to the mimivirus. This implies that there were mimivirii around when we and the amoeba shared an ancestor and that we still possess the code to generate antibodies, although GrrlScientist's question about smallpox raises the possibility that the mimivirus reaction was a convergent general purpose reaction to any large virus.
It strikes me that a fruitful avenue of inquiry on virophages would be to look for one that infests cowpox, which I believe still exists in the wild and, since it is a close smallpox relative, would also be a large virus.
A couple of further thoughts on evolutionary pressures for these three organisms (or however you want to define them):
As I read this we have three organisms:
an amoeba (call it "A" to save typing)
a mimivirus (call it MV)
and a virophage (call it VP).
MV is infectious to A; VP is infectious to MV but only when MV is active inside A. If MV is active in A and infected with VP then A is less likely to lyse (break open, spilling new MV and/or VP particles). It strikes me that this property of VP is counter to evolutionary sense. VP should *not* prevent MV from causing the cell to lyse, as it reduces VPs reproductive potential. If A does not lyse, then no new VP particles are shed (I am guessing that a VP-infected A would not shed active MV particles when it did lyse*). In the instances where A does lyse, then only those VP particles are spread, which should limit any tendency of VP to prevent lysis.
Now in the absence of MV, VP does not reproduce inside A, I understand that, but it would seem to me that there would be a benefit to A if it could either host a minimal load of VP particles (perhaps as organelles) or co-opt the anti-lysis properties of VP. why has it not done so? Especially if those VP particles that are "best" at preventing lysis are those that are most likely to hang around inside A.
*although Raoult and colleagues did discover VP in association with MV, so perhaps they grow and are shed together?
I hope these questions make some limited sense, I am full of thought on how the pressures run in this three-body problem and I hope there are at least a couple of theses in this discovery!
apikoros,
Suppose VP spreads by budding off from the infected A. In that case, VP would want to keep M from lysing A, and also keep from killing A itself, so that the M+VP-infected A can serve indefinitely as a factory for making VP. A, of course, doesn't want either of them around.
Meanwhile, I'm reminded of this by Jonathan Swift:
So, naturalists observe, a flea
Has smaller fleas that on him prey;
And these have smaller still to bite 'em;
And so proceed ad infinitum.
Holy carp. This is one of the coolest things I have ever seen in biology. A virus-infecting virus...
I think instead of updating the definition of life we might have to update the definition of machine. It's like a von Neumann probe living off of bigger von Neumann probes living in a terrain that happens to be living flesh.
Qetzal,
Very possible! Yet another possible PhD thesis topic :-) I'd still maintain that an amoeba that could maintain some VP as an organelle or co-opt the anti-lysis and MV-disabling features would be ahead of the game, so I see selective pressure in that direction, but there may be some counterpressure, it may turn out that there is a strain of amoeba that have done this, or it may just not have happened yet. I've no doubt that this lab will be actively reporting results for quite a while!
Microbiology is my field so this find is especially exciting to me. Thanks so much for sharing and this was a wonderful overview of the paper.
A bit off the point but I can't help thinking - I am always thrilled but never surprised by discoveries which push us to expand our definition of life. Stories like this make it seem all the more possible that, somewhere else in our solar system, life has found a way. I watch Mars with bated breath. With some luck, what we find there will also require us to rethink what life is.
Thanks, qetzal - I was reminded of that too, but couldn't remember the full quote, or the author.
Sod the genetics, I'd love to know more about how exactly Sputnik is parasitising mamavirus. I guess it's wrapping itself in the mamavirus capsid, which is causing the mamavirus to produce deformed mamaviruses. But I'm sure the fuller story is more interesting.
apikoros - what effect does infection by both MV and VP have on the amoeba's fitness? Even if lysis is delayed, the amoeba might still be reproductively dead: it might be too busy producing VP to reproduce itself.
Very interesting.
But being really picky, is it really a virus infecting another virus, or:
a virus co-transinfecting in association with another virus and/or integrating into the virion of the other virus
--?
Hope that makes sense. I like the idea of co-transinfection and also of integrating into an (integrated) virion (think of site-specific integration).
And a virus with 900 protein-encoding genes??! That's a massive virus. Ventor's "smallest genome" has a lot fewer (180+ genes I think).
Bob O'H - I don't know. If both MV and VP prevent normal reproduction, then both are equally bad. If, however VP does not interfere with A's reproduction then VP is conferring a benefit on A (protection from lysis by MV) and I would expect some sort of symbiosis to evolve. Even if initially VP did interfere with A's reproduction, I would propose looking into whether A and VP are co-evolving to reduce the interference. If this were to happen, we might very well be looking at the beginnings of the evolution of antibodies.
My initial question was why VP should delay lysis. From VPs point of view, delaying or preventing lysis would seem to reduce VPs reproductive potential, as those As that do not lyse do not spread VP particles.
I'm sorry to say that I came to this thread via Balloon Juice. I'm not a biologist, but rather a computer programmer specialized in economics. All I have is a head full of ideas and questions, some of which are sure to be ridiculous through my own ignorance.
I am not a biologist, but I have followed the discoveries of various extremophiles with interest. I have listened when respected scientists speculate on the realistic possibilities for life on Titan, on Europa, in the upper atmosphere of Venus, and of course Mars. It seems clear that our definition of life and its boundaries is woefully inadequate and that we will see it undergo significant revisions as science continues to push the boundaries of exploration.
apikoros,
I don't know anything specific about this Sputnik virus. But it's well established that some viruses escape their host via a sort of exocytosis procedure, rather than by lysing the cell.
Thus, it's premature to conclude that VP can't spread if A doesn't lyse. Obviously VP spreads somehow, which suggests one of two possibilities. VP may only delay lysis of A, for its own (currently inscrutable) reasons.
Or, VP may have an alternate means of escaping A and spreading, as observed for other viruses.
Or there could be a third possibility that I haven't even counted upon (cue Alice's Restaurant music).
Maybe someone else mentioned this and I missed it, but this virus phage is similar to AAV. A small incomplete virus that can only replicate in the presence of an Adenovirus infection, though we have modified it to work without adenovirus coninfection for gene therapy purposes. Perhaps what we are seeing here is something along those lines but the smaller defective virus had run amok and is damaging the host virus.
Travis -
I thought of AAV as well. Interestingly, the concluding paragraph of the Nature letter reads as follows:
I'm not a virologist, but the authors seem to imply that Sputnik is not that comparable to AAV.
I haven't had a chance to read the article yet, hopefully I have time this weekend. But to me the mechanism doesn't seem to be that different from how a defective virus propagates, the difference is in the result. AAV could be looked at as parasite that gets along with the host and does little to no harm to it, while this virus is a newly formed parasite that hasn't quite gotten into tune with it's host and is still killing them off. Perhaps we are seeing a stage of the co evolution of a phage and it's host where the phage is kicking the hosts butt? Not to say it isn't interesting, it certainly is.
@apikoros: Possessing antibodies to mimivirus does *not* imply that the virus has been around since the common ancestor of humans and amoebae; it instead implies that a modern human was infected with an amoeba that itself was infected with mimivirus. The mimivirus then lysed its host amoeba cell, releasing releasing the mimivirus virions, at which point a modern human immune system made antibodies against them.