"The atoms come into my brain, dance a dance, and then go out - there are always new atoms, but always doing the same dance, remembering what the dance was yesterday." -Richard Feynman
Here you are, a human being, a grand Universe of atoms that have organized themselves into simple monomers, assembled together into giant macromolecules, which in turn comprise the organelles that make up your cells. And here you are, a collection of around 75 trillion specialized cells, organized in such a way as to make up you.
But at your core, you are still just atoms. A mind-bogglingly large number of atoms -- some 1028 of them -- but atoms nonetheless.
Those two things -- you and an atom -- may seem so different in scale and size that it's hard to wrap your head around. Here's a fun way to think about atoms: if you broke down a human being into all the atoms that make you up, there are about as many atoms that make up you (~1028) as there are "a-human's-worth-of-atoms" to make up the entire Solar System!
 Image credit: Superb Wallpapers, via http://www.desktoplemming.com/.
Image credit: Superb Wallpapers, via http://www.desktoplemming.com/.
All the matter in the Solar System, all summed together, contains about 1057 atoms, or 1029 human-beings-worth of atoms. So an atom, compared to you, is as tiny as you are in comparison to the entire Solar System, combined.
But that's just for perspective. The 1028 atoms that are existing-as-you-right-now each have their own story stretching back to the very birth of the Universe. Each one has its own story, and so today I bring you the story of just one atom in the Universe.
There was a time in the distant past -- some 13.7 billion years ago -- when there were no atoms. Yes, the energy was all there, but it was far too hot and too dense to have even a single atom. Imagine all the matter in the entire Universe, some 1091 particles, in a volume of space about equal to that of a single, giant star.
The whole Universe, compressed into a volume of space that one large star takes up.
Yes, back then it was too hot to have any atoms at all. But the Universe didn't stay that way for long: it may have been incredibly hot and dense, but it was expanding and cooling incredibly rapidly back then. After less than a second, the quarks and gluons had condensed into stable protons and neutrons, the building blocks of all atomic nuclei.
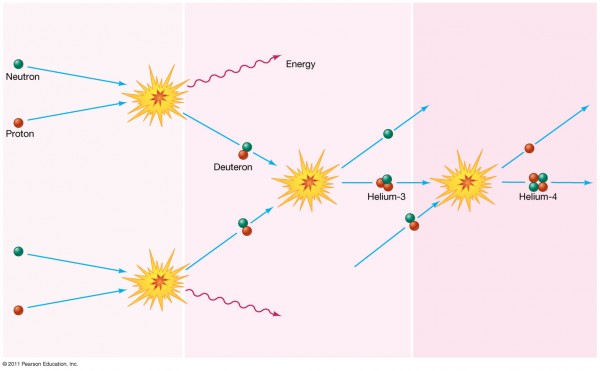
The atom we're thinking of started out as a neutron. Protons tried to fuse with it to create deuterium, but the Universe was too hot for that to happen, and each time it formed deuterium, it was blasted apart less than a nanosecond later.
After about three minutes, a few of the neutrons had decayed into protons, but this one remained, and finally the Universe had cooled enough so that nuclear fusion could proceed. The neutron quickly formed deuterium, then Helium-3, and finally found another deuteron to become a Helium-4 nucleus. Only about 8% of the atoms in the Universe became Helium-4 like this one; the other 92% were just plain old protons, also known as Hydrogen nuclei.
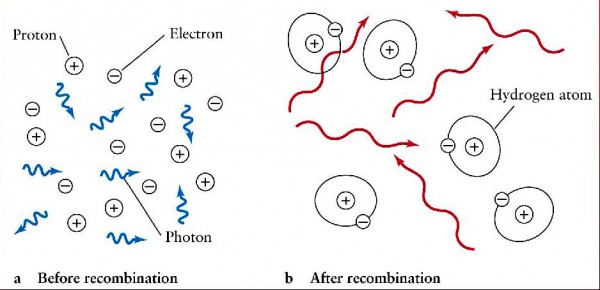
It took another 380,000 years for the Universe to cool enough for this to become a neutral atom, and for two electrons to join this nucleus. The Universe -- despite its rapid expansion and cooling -- remains 100% ionized until the temperature drops to just a few thousands of degrees, which simply takes that much time.
Over the next hundred-million years or so, this atom found itself caught up in the gravitational pull of the Universe, which began to form stars and galaxies. But the vast majority of atoms -- more than 95% -- weren't a part of the first generation of stars, and neither was this one in particular.
Instead, when the first stars formed, they kicked the electrons out of the atoms that surrounded them, creating ions once again.
It was only by luck that this atom we're following wound up in a dense molecular cloud, shielded from this radiation. After more than a billion years in this collection of neutral atoms, it finally found itself pulled in by gravitational attraction to what would become a giant star.
This atom lost its electrons and fell to the core of the star, where it lay dormant for millions of years, as hydrogen nuclei fused into other helium nuclei just like this one. When the core ran out of hydrogen fuel, helium fusion began, and our atom fused with two others to become a carbon nucleus!
While other atoms even closer to the center of the star fused further, carbon was as far as this particular atom went. When the core of the star collapsed and the star went supernova, our atom was blown out into the interstellar medium, where it resided for billions of years.
 Image credit: X-ray: NASA/CXC/Caltech/S.Kulkarni et al.; Optical: NASA/STScI/UIUC/Y.H.Chu & R.Williams et al.; IR: NASA/JPL-Caltech/R.Gehrz et al.
Image credit: X-ray: NASA/CXC/Caltech/S.Kulkarni et al.; Optical: NASA/STScI/UIUC/Y.H.Chu & R.Williams et al.; IR: NASA/JPL-Caltech/R.Gehrz et al.
While billions of other stars went through the life-and-death cycle, this carbon atom remained in interstellar space, eventually picking up six electrons to become neutral. It found its way into a gravitational collection of neutral gas, and cooled, eventually getting sucked in to another gravitational perturbation, as star-formation happened all over again.
 Image credit: Tony Hallas of http://www.astrophoto.com/.
Image credit: Tony Hallas of http://www.astrophoto.com/.
This time, the atom didn't find its way into the central star of its system, but rather into the dusty disk that surrounded it. Over time, the disk separated into planetoids and planetesimals, and this atom found itself aboard one of those.
It first joined together with four hydrogen atoms, becoming methane, and went through millions of different chemical reactions over time.
 Image credit: 2009, Zappa, via http://macroworlds.com/.
Image credit: 2009, Zappa, via http://macroworlds.com/.
After life took hold on Earth, it became a part of a bacterium's DNA, then a part of a plant's cell wall, and eventually became part of a complex organism that would find itself consumed by you.
 Image credit: Anna of http://mtkilimonjaro.blogspot.com/.
Image credit: Anna of http://mtkilimonjaro.blogspot.com/.
The atom is currently in a red blood cell of yours, where it will remain for a total of about 120 days, until the cell is destroyed and replaced by a different one.
Although the cell -- and all cells in your body -- will be destroyed and replaced, you will remain the same person you are, and the atom will simply take on a different function, whether in your body or out of it. The atoms in your body are temporary, and can all be replaced -- unnoticed by you -- by another of the same type.
 Image credit: Youngester of http://technicalstudies.youngester.com/
Image credit: Youngester of http://technicalstudies.youngester.com/
And each of the 1028 atoms in your body has a story as spectacular and unique as this one! As Feynman famously said,
"I / a Universe of atoms / an atom in the Universe."
The story of the Universe is inside every atom in your body, each and every one. And after 13.7 billion years, 10,000,000,000,000,000,000,000,000,000 of them have come together, and that's you. The Universe is inside of you, as surely as you're inside the Universe.
You, a Universe of atoms, an atom in this Universe.







I want to hear the one about the atoms then end up in my beer!
Mmmmm, atoms.
Well, one epoch a carbon atom went on a bender with a helium atom and came out oxygen, but it doesn't remember much of what happened after that.
I love science that is told so I can understand it and appreciate it best thing I have ever seen written thankyou
Another great post!
Best article EVER!
Ethan, I love your articles as they so often fill in gaps in my knowledge about the universe. But this article highlighted one thing that I realize I don't understand. If the big bang originated from a point in space and at one instant in time had the energy density equivalent to 10^91 atoms in the volume of a large star, why didn't gravitation result in a hypermassive black hole and prevent expansion of the matter/energy? Is this a mystery that can only be explained once we have a Theory of Everything with Quantum Gravity, or is there an accepted explanation using contemporary Cosmology that I simply haven't come across? (Or maybe I have come across it, but it wasn't explained with the clarity that you regularly achieve, and therefore failed to fully grasp it.)
I'm with Jim above. I've always wondered how the universe managed to expand out of a singularity. Also, has all the mass of the universe always existed, or has it increased since the Big Bang through things like the Casimir effect?
"If the big bang originated from a point in space "
And if it didn't?
Because you're using monkey-bananas-want-language (which is any human language developed to tell another monkey where the bananas were) to produce a sentence that is almost entirely wrong.
At the big bang, all points were at that spot. It wasn't one spot. It was ALL OF THEM.
So, what if your description is not what's going on?
Interesting to think about the first atom.
The Adam Atom, you might say.
@Wow: ok, thanks for pointing out my sloppy wording. I should have said something like "If space had near infinitesimal volume at an instant after the big bang." But this is a distraction, as it is irrelevant to my question. Ethan said "Imagine all the matter in the entire Universe, some 10^91 particles, in a volume of space about equal to that of a single, giant star." That would seem to imply the conditions for a black hole. What am I missing?
I have read Brian Greene's books so I am aware that one hypothesis from M-Theory is that the big bang resulted from the collision of two membranes, in which case the Big Bang could have happened over a very large volume of 3-dimensional space, releasing a vast amount of energy distributed over that space without concentrating so much energy in any given volume to cause gravitational collapse into a black hole. But this is not the scenario that Ethan implies in the article.
"That would seem to imply the conditions for a black hole. What am I missing?"
Time. It still takes time for the gravitational field from one end of the volume to "see" the gravitational field from the other. So the mass isn't all in one clump like it would be in an old universe which allows black holes.
Inflation. The universe was expanding faster than it could fall back because of the lack of the-rest-of-the-universe-being-noticed.
To an extent the entire universe is a black hole to something "outside".
And several other things which you'd need postgraduate education and a good long run-up to understand. And I can't be bothered to run at my age :-)
@Wow: "Time. It still takes time for the gravitational field from one end of the volume to “see” the gravitational field from the other."
Ah, that helps a great deal. So gravity as a force was active, but with space inflating so rapidly gravity effectively had no effect.
Thanks!
No worries.
Sorry, folks, I'm not buying it. I read your fantastic tale of how it all happened, but I didn't hear any attempt to explain how you know it happened this way. Did you use the scientific method? Tell me which of the "facts" you listed have been proved and which are mere guesses. And what "facts" were used to determine how and when all this came about. Until you can prove any of this, it amounts to metaphysics--not science.